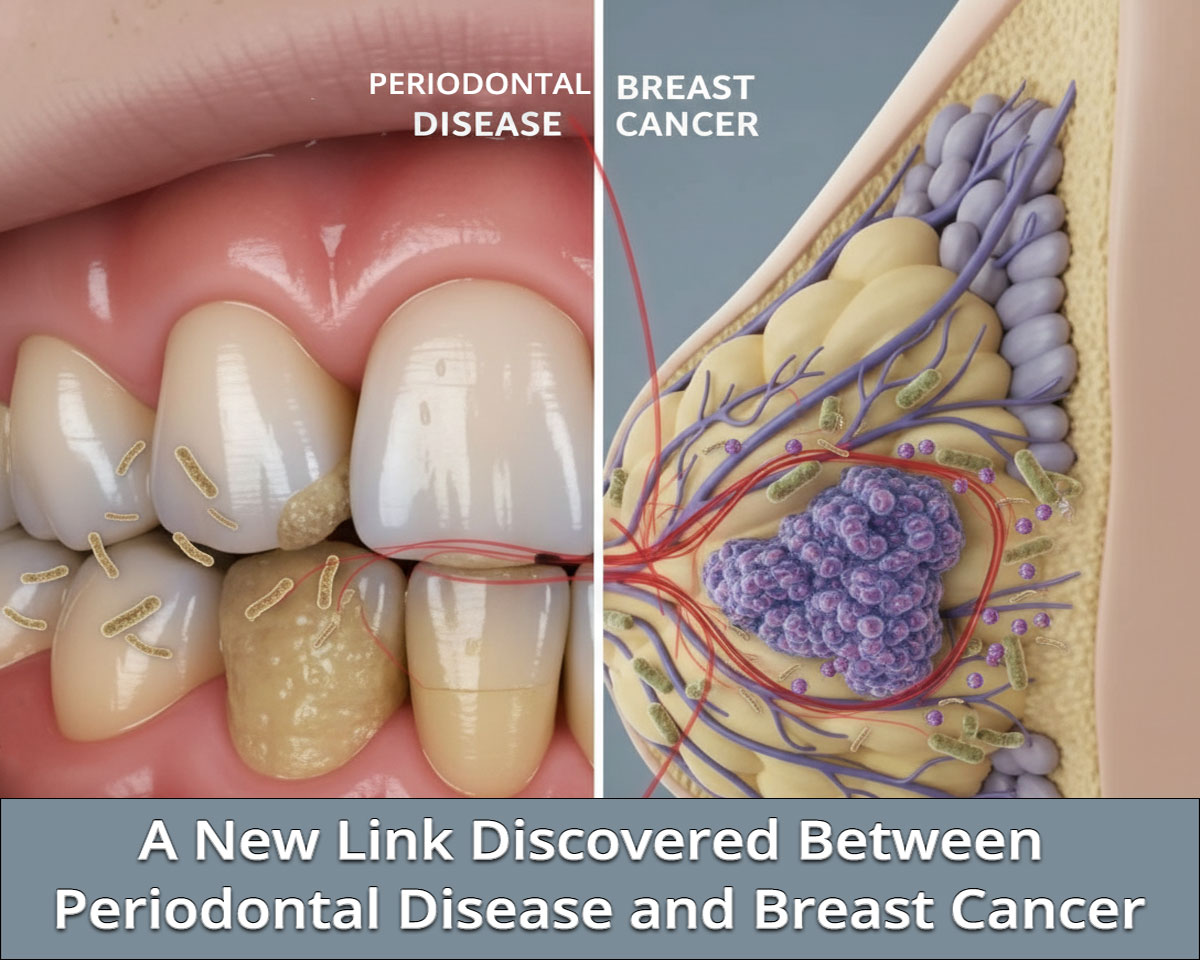
A New Link Discovered Between Periodontal Disease and Breast Cancer
Emerging evidence is redefining how oral health intersects with systemic disease. A growing body of research now suggests that periodontal pathogens—particularly Fusobacterium nucleatum—may contribute to breast cancer initiation, progression, and treatment resistance. For dental professionals, this evolving science reinforces the importance of periodontal disease management as part of broader systemic risk reduction.
This article reviews the latest mechanistic and translational evidence connecting periodontal disease, microbial dysbiosis, and breast cancer, with particular focus on genetically susceptible populations such as BRCA1 mutation carriers.
Breast Cancer Risk: Beyond Genetics and Lifestyle
Breast cancer remains one of the most prevalent malignancies worldwide. While established risk factors include age, hormonal exposure, obesity, alcohol use, and genetic predisposition, approximately 30–45% of women carrying pathogenic BRCA1 mutations never develop breast cancer. This variability suggests that environmental or microbial co-factors may modulate cancer risk in genetically susceptible individuals.
Recent research has shifted attention to the human microbiome as a potential modifier of carcinogenesis. Distinct microbial signatures have been identified in breast tumors compared to adjacent normal tissue, and dysbiosis has been associated with tumor aggressiveness and metastatic behavior.
The oral microbiome—particularly in the setting of chronic periodontal disease—has emerged as a potential contributor.
Fusobacterium nucleatum: From Periodontal Pathogen to Oncogenic Driver
Fusobacterium nucleatum is a gram-negative, anaerobic bacterium commonly found in dental biofilm. It plays a well-established role in the pathogenesis of periodontitis through biofilm formation, inflammatory activation, and tissue destruction.
However, beyond the oral cavity, F. nucleatum has been implicated in multiple malignancies, including:
Colorectal cancer
Pancreatic cancer
Head and neck cancers
Oral squamous cell carcinoma
Now, mounting evidence indicates that this periodontal pathogen may also inhabit breast tumor tissue.
Metagenomic analyses of breast cancer samples have demonstrated overabundance of F. nucleatum and associated oral biofilm-forming bacteria in both benign and malignant breast tissue. Notably, the abundance of these pathogens in breast tumors positively correlates with their oral levels in the same patient—suggesting possible translocation from the oral cavity.
How Does an Oral Bacterium Reach the Breast?
Several mechanisms have been proposed:
Hematogenous dissemination during bacteremia episodes
Mammary-intestinal microbial axis
Direct nipple duct colonization
Transient bacteremia is common in patients with active periodontal disease, particularly during routine oral activities such as brushing or mastication. This provides a plausible biological route for systemic microbial dissemination.
Experimental models support this hypothesis.
Intraductal Colonization Triggers Pre-Cancerous Changes
In mouse models, direct colonization of mammary ducts with F. nucleatum resulted in:
Metaplastic and hyperplastic lesions
Marked inflammatory infiltration
Increased collagen deposition and fibrosis
DNA damage markers
Hyperproliferation of mammary epithelial cells
Importantly, these changes developed in otherwise healthy mice within three weeks of exposure.
These findings suggest that mammary duct colonization by periodontal pathogens may be sufficient to initiate early pathologic changes consistent with tumorigenic transformation.
Systemic Exposure Accelerates Tumor Growth and Metastasis
When breast tumor-bearing mice were exposed to F. nucleatum via hematogenous administration:
Tumor growth significantly accelerated
Tumor volume and weight increased
Lung metastases occurred in 100% of exposed animals (vs. 20% in controls)
Tumors demonstrated increased proliferation markers (Ki67, PCNA)
Gene expression analysis revealed activation of oncogenic pathways
Key enriched pathways included:
Wnt/β-catenin
KRAS signaling
mTOR
Myc activation
Epithelial-to-mesenchymal transition (EMT)
Functionally, tumor cells exposed to F. nucleatum exhibited:
Increased migration
Enhanced invasion
Greater stemness characteristics
Increased mammosphere formation
These are hallmarks of aggressive and metastatic cancer behavior.
DNA Damage and Error-Prone Repair: A Critical Mechanism
One of the most significant findings relates to DNA damage response.
Exposure to F. nucleatum induced:
γ-H2AX foci formation (a marker of double-strand DNA breaks)
Activation of ATM kinase signaling
Upregulation of DNA-dependent protein kinase catalytic subunit (DNA-PKcs)
Engagement of the non-homologous end joining (NHEJ) pathway
NHEJ is an error-prone DNA repair mechanism. While it restores DNA integrity, it increases the likelihood of mutational accumulation.
Overexpression of DNA-PKcs has been associated with:
Cancer progression
Chemotherapy resistance
Radiation resistance
Thus, periodontal pathogen exposure may not only promote tumor growth but also enhance genomic instability.
BRCA1 Mutation: A Vulnerable Population
Perhaps the most clinically relevant discovery involves BRCA1-mutant breast epithelial cells.
F. nucleatum binds to host cells through interaction between its Fap2 adhesin protein and the Gal-GalNAc sugar residue expressed on the surface of transformed cells.
Research shows:
BRCA1-mutant breast epithelial cells exhibit significantly higher surface accumulation of Gal-GalNAc
These cells internalize and retain F. nucleatum more efficiently
Bacterial retention persists across cellular generations
Even brief exposure (4 hours) induces sustained biological effects
This suggests that BRCA1 mutation carriers may be biologically more susceptible to colonization and oncogenic effects of periodontal pathogens.
Given that BRCA1 mutations alone are insufficient to cause cancer in all carriers, microbial dysbiosis may represent a critical “second hit.”
Increased Stemness and Reduced Drug Sensitivity
Breast cancer cells and BRCA1-mutant epithelial cells exposed to F. nucleatum demonstrated:
Increased clonogenicity
Greater anchorage-independent growth
Enhanced migration
Elevated stemness markers (Oct4, Nanog, ALDH1)
Reduced sensitivity to Olaparib (PARP inhibitor)
Reduced sensitivity to Doxorubicin
Chemoresistance is a major driver of poor outcomes in triple-negative breast cancer and BRCA-mutated cancers. These findings suggest that intratumoral bacterial colonization may reduce therapeutic efficacy.
Clinical Implications for Dental Professionals
The emerging evidence positions periodontal disease as a potential modifiable systemic risk factor for breast cancer progression.
For dental professionals, this has significant implications:
1. Periodontal Disease Management as Systemic Prevention
Aggressive control of periodontal inflammation may reduce systemic microbial dissemination.
Key strategies include:
Early diagnosis of periodontitis
Comprehensive scaling and root planing
Maintenance therapy protocols
Biofilm control education
Adjunctive antimicrobial therapies when indicated
2. Interdisciplinary Communication
Patients with:
BRCA1 mutations
Strong family history of breast cancer
Triple-negative breast cancer
Ongoing chemotherapy
may benefit from coordinated periodontal management.
Oncology-dental collaboration is increasingly relevant.
3. Pre-Treatment Oral Health Optimization
For patients undergoing cancer therapy:
Elimination of active periodontal infection
Stabilization of inflammatory burden
Prevention of bacteremia events
may have implications beyond oral health alone.
Periodontal Disease: A Potential Modifiable Risk Factor
While causality in humans remains under investigation, the mechanistic evidence is compelling:
Oral pathogens detected in breast tumors
Direct colonization induces precancerous changes
Hematogenous exposure accelerates metastasis
DNA damage and genomic instability are triggered
BRCA1-mutant cells show heightened susceptibility
These findings strengthen the systemic model of oral infection and chronic inflammation.
The Future of Oral-Systemic Oncology Research
Further human cohort studies are needed to clarify:
Whether periodontal therapy reduces breast cancer risk
Whether oral microbial profiling predicts oncologic outcomes
Whether antimicrobial strategies improve chemotherapy response
However, the biological plausibility is now well established.
For dental professionals, the message is clear: periodontal health extends far beyond the gingiva.
Conclusion
The discovery of Fusobacterium nucleatum in breast tumor tissue provides a compelling new link between periodontal disease and breast cancer biology. Evidence demonstrates that this oral pathogen can:
Induce metaplastic lesions in healthy mammary tissue
Accelerate tumor growth and metastasis
Trigger DNA damage and error-prone repair
Promote stemness and chemoresistance
Preferentially target BRCA1-mutant cells
As research continues to unravel the oral-systemic axis, periodontal disease management may become an integral component of comprehensive cancer risk reduction strategies.
For dental professionals, this represents both a responsibility and an opportunity: protecting periodontal health may ultimately contribute to protecting systemic health—including the breast.
Reference:
A pro-carcinogenic oral microbe internalized by breast cancer cells promotes mammary tumorigenesis
Published:
Recent Articles
Emerging evidence is redefining how oral health intersects with systemic disease. A growing body of research now suggests that periodontal pathogens—particularly Fusobacterium nucleatum—may contribute to breast cancer initiation, progression, and treatment resistance. For dental professionals, this evolving science reinforces the importance of periodontal disease management as part of broader systemic risk reduction.
This article reviews the latest mechanistic and translational evidence connecting periodontal disease, microbial dysbiosis, and breast cancer, with particular focus on genetically susceptible populations such as BRCA1 mutation carriers.
Breast Cancer Risk: Beyond Genetics and Lifestyle
Breast cancer remains one of the most prevalent malignancies worldwide. While established risk factors include age, hormonal exposure, obesity, alcohol use, and genetic predisposition, approximately 30–45% of women carrying pathogenic BRCA1 mutations never develop breast cancer. This variability suggests that environmental or microbial co-factors may modulate cancer risk in genetically susceptible individuals.
Recent research has shifted attention to the human microbiome as a potential modifier of carcinogenesis. Distinct microbial signatures have been identified in breast tumors compared to adjacent normal tissue, and dysbiosis has been associated with tumor aggressiveness and metastatic behavior.
The oral microbiome—particularly in the setting of chronic periodontal disease—has emerged as a potential contributor.
Fusobacterium nucleatum: From Periodontal Pathogen to Oncogenic Driver
Fusobacterium nucleatum is a gram-negative, anaerobic bacterium commonly found in dental biofilm. It plays a well-established role in the pathogenesis of periodontitis through biofilm formation, inflammatory activation, and tissue destruction.
However, beyond the oral cavity, F. nucleatum has been implicated in multiple malignancies, including:
Colorectal cancer
Pancreatic cancer
Head and neck cancers
Oral squamous cell carcinoma
Now, mounting evidence indicates that this periodontal pathogen may also inhabit breast tumor tissue.
Metagenomic analyses of breast cancer samples have demonstrated overabundance of F. nucleatum and associated oral biofilm-forming bacteria in both benign and malignant breast tissue. Notably, the abundance of these pathogens in breast tumors positively correlates with their oral levels in the same patient—suggesting possible translocation from the oral cavity.
How Does an Oral Bacterium Reach the Breast?
Several mechanisms have been proposed:
Hematogenous dissemination during bacteremia episodes
Mammary-intestinal microbial axis
Direct nipple duct colonization
Transient bacteremia is common in patients with active periodontal disease, particularly during routine oral activities such as brushing or mastication. This provides a plausible biological route for systemic microbial dissemination.
Experimental models support this hypothesis.
Intraductal Colonization Triggers Pre-Cancerous Changes
In mouse models, direct colonization of mammary ducts with F. nucleatum resulted in:
Metaplastic and hyperplastic lesions
Marked inflammatory infiltration
Increased collagen deposition and fibrosis
DNA damage markers
Hyperproliferation of mammary epithelial cells
Importantly, these changes developed in otherwise healthy mice within three weeks of exposure.
These findings suggest that mammary duct colonization by periodontal pathogens may be sufficient to initiate early pathologic changes consistent with tumorigenic transformation.
Systemic Exposure Accelerates Tumor Growth and Metastasis
When breast tumor-bearing mice were exposed to F. nucleatum via hematogenous administration:
Tumor growth significantly accelerated
Tumor volume and weight increased
Lung metastases occurred in 100% of exposed animals (vs. 20% in controls)
Tumors demonstrated increased proliferation markers (Ki67, PCNA)
Gene expression analysis revealed activation of oncogenic pathways
Key enriched pathways included:
Wnt/β-catenin
KRAS signaling
mTOR
Myc activation
Epithelial-to-mesenchymal transition (EMT)
Functionally, tumor cells exposed to F. nucleatum exhibited:
Increased migration
Enhanced invasion
Greater stemness characteristics
Increased mammosphere formation
These are hallmarks of aggressive and metastatic cancer behavior.
DNA Damage and Error-Prone Repair: A Critical Mechanism
One of the most significant findings relates to DNA damage response.
Exposure to F. nucleatum induced:
γ-H2AX foci formation (a marker of double-strand DNA breaks)
Activation of ATM kinase signaling
Upregulation of DNA-dependent protein kinase catalytic subunit (DNA-PKcs)
Engagement of the non-homologous end joining (NHEJ) pathway
NHEJ is an error-prone DNA repair mechanism. While it restores DNA integrity, it increases the likelihood of mutational accumulation.
Overexpression of DNA-PKcs has been associated with:
Cancer progression
Chemotherapy resistance
Radiation resistance
Thus, periodontal pathogen exposure may not only promote tumor growth but also enhance genomic instability.
BRCA1 Mutation: A Vulnerable Population
Perhaps the most clinically relevant discovery involves BRCA1-mutant breast epithelial cells.
F. nucleatum binds to host cells through interaction between its Fap2 adhesin protein and the Gal-GalNAc sugar residue expressed on the surface of transformed cells.
Research shows:
BRCA1-mutant breast epithelial cells exhibit significantly higher surface accumulation of Gal-GalNAc
These cells internalize and retain F. nucleatum more efficiently
Bacterial retention persists across cellular generations
Even brief exposure (4 hours) induces sustained biological effects
This suggests that BRCA1 mutation carriers may be biologically more susceptible to colonization and oncogenic effects of periodontal pathogens.
Given that BRCA1 mutations alone are insufficient to cause cancer in all carriers, microbial dysbiosis may represent a critical “second hit.”
Increased Stemness and Reduced Drug Sensitivity
Breast cancer cells and BRCA1-mutant epithelial cells exposed to F. nucleatum demonstrated:
Increased clonogenicity
Greater anchorage-independent growth
Enhanced migration
Elevated stemness markers (Oct4, Nanog, ALDH1)
Reduced sensitivity to Olaparib (PARP inhibitor)
Reduced sensitivity to Doxorubicin
Chemoresistance is a major driver of poor outcomes in triple-negative breast cancer and BRCA-mutated cancers. These findings suggest that intratumoral bacterial colonization may reduce therapeutic efficacy.
Clinical Implications for Dental Professionals
The emerging evidence positions periodontal disease as a potential modifiable systemic risk factor for breast cancer progression.
For dental professionals, this has significant implications:
1. Periodontal Disease Management as Systemic Prevention
Aggressive control of periodontal inflammation may reduce systemic microbial dissemination.
Key strategies include:
Early diagnosis of periodontitis
Comprehensive scaling and root planing
Maintenance therapy protocols
Biofilm control education
Adjunctive antimicrobial therapies when indicated
2. Interdisciplinary Communication
Patients with:
BRCA1 mutations
Strong family history of breast cancer
Triple-negative breast cancer
Ongoing chemotherapy
may benefit from coordinated periodontal management.
Oncology-dental collaboration is increasingly relevant.
3. Pre-Treatment Oral Health Optimization
For patients undergoing cancer therapy:
Elimination of active periodontal infection
Stabilization of inflammatory burden
Prevention of bacteremia events
may have implications beyond oral health alone.
Periodontal Disease: A Potential Modifiable Risk Factor
While causality in humans remains under investigation, the mechanistic evidence is compelling:
Oral pathogens detected in breast tumors
Direct colonization induces precancerous changes
Hematogenous exposure accelerates metastasis
DNA damage and genomic instability are triggered
BRCA1-mutant cells show heightened susceptibility
These findings strengthen the systemic model of oral infection and chronic inflammation.
The Future of Oral-Systemic Oncology Research
Further human cohort studies are needed to clarify:
Whether periodontal therapy reduces breast cancer risk
Whether oral microbial profiling predicts oncologic outcomes
Whether antimicrobial strategies improve chemotherapy response
However, the biological plausibility is now well established.
For dental professionals, the message is clear: periodontal health extends far beyond the gingiva.
Conclusion
The discovery of Fusobacterium nucleatum in breast tumor tissue provides a compelling new link between periodontal disease and breast cancer biology. Evidence demonstrates that this oral pathogen can:
Induce metaplastic lesions in healthy mammary tissue
Accelerate tumor growth and metastasis
Trigger DNA damage and error-prone repair
Promote stemness and chemoresistance
Preferentially target BRCA1-mutant cells
As research continues to unravel the oral-systemic axis, periodontal disease management may become an integral component of comprehensive cancer risk reduction strategies.
For dental professionals, this represents both a responsibility and an opportunity: protecting periodontal health may ultimately contribute to protecting systemic health—including the breast.
Reference:
A pro-carcinogenic oral microbe internalized by breast cancer cells promotes mammary tumorigenesis
Published:
Recent Articles
Magnetically Guided Dental Nanobots (CalBots) Are Real
Magnetically Guided Dental Nanobots (CalBots) Are Real The concept of nanobots in dentistry has long been associated with futuristic speculation. However, recent peer-reviewed research [...]
Peri-Implantitis Treatment & Prevention Methods
Peri-Implantitis Treatment & Prevention Methods Peri-implantitis remains one of the most significant biological complications affecting dental implants, posing a serious threat to long-term implant [...]
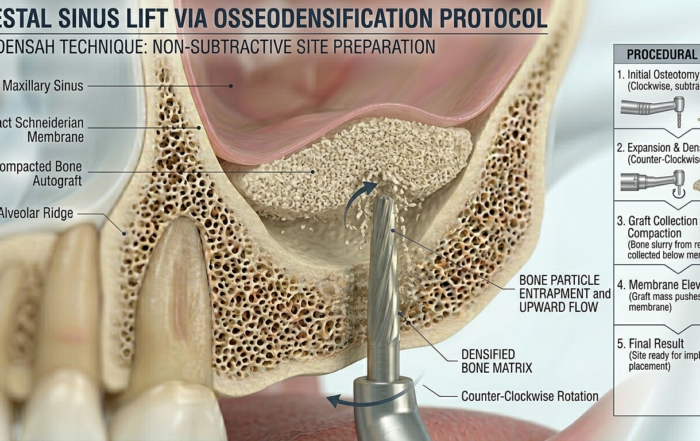
Latest Sinus Lifting Techniques in Modern Implant Dentistry
Latest Sinus Lifting Techniques in Modern Implant Dentistry A Comprehensive Clinical Review Introduction to Sinus Lifting in Implantology Sinus lifting, or maxillary sinus floor [...]
Biomechanics of the Triple Abutment & BOPiT Concept
Dental Biomechanics · Implant Science · Clinical Evidence Biomechanics of the Triple Abutment & BOPiT Concept How a saddle-shaped mathematical surface is rewriting the rules of load distribution [...]
The Woman Who Proved One Implant Could Hold Three Crowns
The Woman Who Proved One Implant Could Hold Three Crowns While the dental establishment looked away, Dr. Luciana Colepícolo spent 12 years building the [...]
ENPP1, The Molecular Brake That Limits Tissue Healing
ENPP1, The Molecular Brake That Limits Tissue Healing Introduction: A New Biological Barrier to Dental Regeneration Meet ENPP1—a protein most dentists have never heard [...]