Latest Extraction Socket Regeneration Techniques
Introduction
The alveolar bone surrounding a tooth is a highly specialized structure characterized by a distinctive microarchitecture. One of its defining features is the dual orientation of collagen fibers created by the insertion of Sharpey’s fibers into the bone matrix. In addition, the alveolar bone is richly vascularized and innervated, with numerous blood vessels and nerve fibers penetrating the structure. Because teeth are constantly subjected to functional loading and biomechanical forces, the surrounding bone undergoes continuous remodeling to maintain structural integrity and adapt to mechanical demands.
The morphology and volume of the alveolar bone are closely influenced by the characteristics of the associated tooth, including its inclination, eruption axis, and anatomical form. When a tooth is extracted, the surrounding alveolar socket inevitably undergoes a cascade of biological and structural changes. These post-extraction events lead to measurable alterations in both the vertical and horizontal dimensions of the alveolar ridge. As a consequence, the available bone volume may be significantly reduced over time.
Such dimensional changes can complicate subsequent implant therapy. Insufficient bone width or height can make implant placement technically challenging, compromise primary stability, and potentially affect osseointegration. Furthermore, loss of alveolar bone volume may negatively influence the esthetic outcome of prosthodontic rehabilitation, whether through implant-supported restorations or conventional prostheses.
Clinical evidence highlights the magnitude of this post-extraction remodeling. A systematic review by Niklaus P. Lang and colleagues quantified the dimensional alterations occurring after tooth removal. Their analysis demonstrated that within six months following extraction, horizontal bone loss typically ranges between approximately 2.5 mm and 4.5 mm, while vertical loss averages around 0.8–1.5 mm. The buccal plate tends to experience more pronounced resorption compared with lingual or palatal surfaces, and buccolingual shrinkage is usually greater than mesiodistal reduction. Importantly, the majority of this bone loss occurs during the first three to six months after extraction, after which the rate of resorption gradually slows.
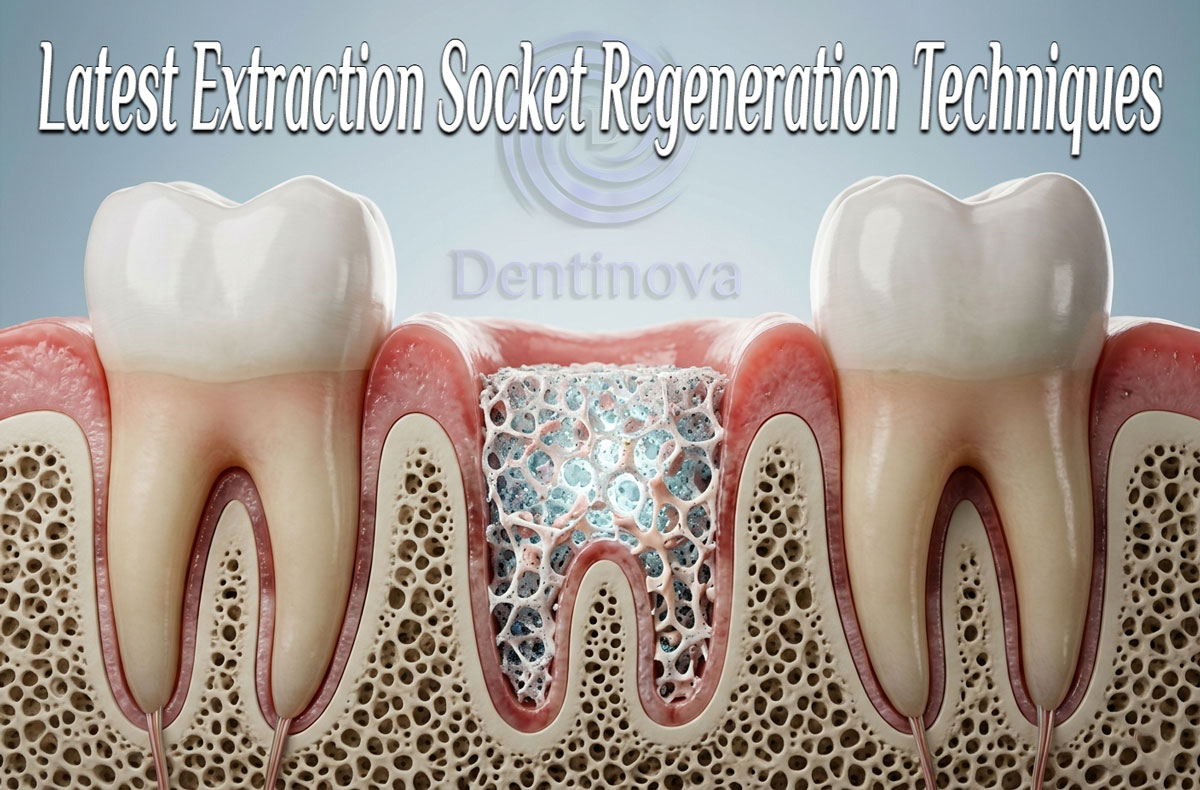
To counteract these dimensional changes, socket preservation procedures have been developed to maintain the structural integrity of the alveolar ridge. These interventions aim to limit early bone resorption and preserve ridge volume immediately after tooth removal. According to the definition proposed by Daniel S. Thoma Hammerle and colleagues, ridge preservation refers to maintaining the existing ridge dimensions within the envelope present at the time of extraction.
Clinical indications for ridge preservation include cases in which immediate or early implant placement is not feasible, when primary implant stability cannot be achieved, or when prosthodontic treatment requires preservation of ridge contour. With the rapid growth of implant dentistry, ridge preservation is increasingly considered an essential step following extraction in many clinical scenarios. By maintaining both hard and soft tissue architecture, socket preservation simplifies future implant placement and improves functional and esthetic outcomes.
The purpose of this review is to outline the most current extraction socket regeneration techniques and to provide dental professionals and implantologists with an evidence-based overview of modern alveolar ridge preservation strategies.
Healing of the Extraction Socket
A comprehensive understanding of extraction socket healing is fundamental for clinicians seeking to implement effective regeneration or preservation techniques. The healing process follows a well-defined sequence of biological events that involve both resorptive and regenerative mechanisms.
Immediately after tooth extraction, the socket becomes filled with blood, forming a coagulum. This blood clot acts as a temporary scaffold and is subsequently replaced by a provisional matrix composed of fibrin and inflammatory cells. The provisional matrix supports cellular migration and provides the initial framework for tissue regeneration.
During the early stages of healing, osteogenic cells begin to populate the socket, leading to the formation of woven bone. This immature bone tissue gradually bridges the socket and provides the first hard tissue structure within the extraction site. Woven bone is characterized by a disorganized collagen framework and relatively low mechanical strength.
As healing progresses, the remodeling phase begins. During this stage, osteoclastic activity resorbs the immature woven bone while osteoblastic cells deposit more organized lamellar bone. Successive layers of lamellar bone strengthen the newly formed hard tissue bridge and contribute to the maturation of the socket structure.
Eventually, the cortical bone plate begins to develop along the outer boundaries of the socket. Simultaneously, the periosteum forms, and the overlying mucosal tissues attach to the cortical surface. This process, in which bone remodeling results in the development of a cortical wall around the socket, is known as corticalization.
The sequence of clot formation, woven bone deposition, and lamellar bone maturation closely resembles the healing process observed in long-bone fractures. Understanding this physiological progression allows clinicians to design regenerative strategies that enhance osteogenesis while minimizing alveolar ridge resorption.
Socket Preservation Techniques
Multiple techniques and biomaterials have been proposed to preserve the alveolar ridge following tooth extraction. Over the past few decades, advances in biomaterials, molecular biology, and regenerative dentistry have significantly expanded the available therapeutic options.
Socket preservation strategies now range from minimally invasive surgical approaches, such as flapless extraction, to sophisticated regenerative procedures involving biomaterials, biologic mediators, and cell-based therapies. These techniques aim to stabilize the extraction socket, promote bone regeneration, and limit the natural resorptive processes that occur after tooth removal.
Modern alveolar ridge preservation approaches include immediate implant placement, bone grafting, barrier membrane application, platelet-derived biologics, growth factor therapies, tooth-derived graft materials, and emerging tissue engineering techniques involving stem cells.
3.1 Immediate Implants as Socket Preservers
Traditionally, dental implant placement is delayed for approximately six to twelve months following tooth extraction to allow complete healing of the socket. However, during this healing period, substantial resorption of the alveolar ridge may occur, potentially complicating later implant placement and compromising treatment outcomes.
Immediate implant placement has been proposed as a strategy to reduce post-extraction bone resorption by occupying the socket immediately after extraction. The rationale is that the implant fixture may help maintain alveolar ridge dimensions and minimize bone remodeling.
Some clinicians have advocated immediate implant placement without additional preservation procedures such as barrier membranes or graft materials. This approach was suggested as a means to prevent dimensional changes in the alveolar ridge while simultaneously shortening treatment time.
However, clinical and experimental studies have reported mixed outcomes regarding the effectiveness of immediate implants in preventing bone loss. Research conducted by Marcelo Araujo demonstrated that even when implants were placed immediately after extraction, measurable bone loss still occurred during the healing phase. In experimental models, approximately 2.6 mm of bone reduction was observed after three months of healing.
Additional investigations showed that while initial bone-to-implant contact could be established within one month, resorption of the buccal plate often occurred subsequently. As the buccal bone wall resorbed, some of the previously established bone-to-implant contact was partially lost during later stages of healing.
Clinical research conducted by Tiziano Botticelli further confirmed that immediate implant placement does not completely prevent the physiological bone remodeling that occurs after extraction. Even when implants were immediately loaded, the extent of buccal bone resorption remained largely unchanged.
These findings indicate that although immediate implants offer advantages such as reduced treatment time and fewer surgical interventions, they do not fully eliminate post-extraction bone resorption. As a result, clinicians often combine immediate implantation with additional ridge preservation techniques to improve outcomes.
3.2 Bone Grafts and Bone Substitutes
Bone grafting remains one of the most widely used approaches for extraction socket regeneration. These materials function primarily as scaffolds that support new bone formation while limiting the collapse of the socket walls.
Autogenous Bone Grafts
Autogenous bone grafts are harvested directly from the patient and are widely regarded as the gold standard in bone regeneration. Common intraoral donor sites include the maxillary tuberosity, mandibular ramus, mandibular symphysis, and edentulous ridges. Extraoral donor sites such as the iliac crest or tibia may also be used when larger volumes are required.
Autogenous grafts possess osteoconductive, osteoinductive, and osteogenic properties. They support bone formation by providing living cells, growth factors, and a structural scaffold. Additionally, they are biocompatible and do not provoke immunological reactions.
Allogenic Bone Grafts
Allogenic grafts, or allografts, are obtained from donors of the same species. These grafts are typically processed through techniques such as freeze-drying or demineralization to ensure safety and reduce antigenicity.
Demineralized freeze-dried bone allograft (DFDBA) is particularly notable for its osteoinductive potential because the demineralization process exposes bone morphogenetic proteins. However, clinicians must consider the theoretical risk of disease transmission and immune response associated with donor-derived materials.
Xenografts and Alloplastic Materials
Bone substitutes also include xenografts and alloplastic materials. Xenografts originate from different species, commonly bovine, porcine, or equine, and function primarily as osteoconductive scaffolds.
Alloplastic materials are synthetic bone substitutes manufactured under controlled laboratory conditions. These materials include bioceramics such as hydroxyapatite and biphasic calcium phosphate, as well as biodegradable polymers. Because they are synthetically produced, their structural properties, including porosity and degradation rate, can be precisely controlled.
Numerous studies have demonstrated that these materials can effectively reduce alveolar ridge resorption when used as socket fillers, although the degree of new bone formation varies depending on the biomaterial used.
3.3 Barrier Membranes in Socket Preservation
The concept of barrier membranes in regenerative dentistry originates from the principle that different cell populations compete to colonize healing wounds. According to a hypothesis proposed by Melvin Melcher, the migration of non-osteogenic cells, such as gingival epithelial cells, into a bone defect may inhibit bone regeneration by preventing osteogenic cells from populating the site.
To address this issue, barrier membranes were introduced to selectively exclude undesirable cell types and create a protected environment for bone regeneration. This concept led to the development of guided tissue regeneration (GTR), later adapted into guided bone regeneration (GBR) for implant dentistry.
In GBR procedures, a membrane is placed over the extraction socket to isolate the defect and maintain space for osteoprogenitor cells. By preventing soft tissue invasion, the membrane allows osteogenic cells to proliferate and differentiate, thereby enhancing new bone formation.
Barrier membranes are broadly categorized into non-resorbable and resorbable types.
Non-resorbable membranes include materials such as expanded polytetrafluoroethylene (ePTFE) and titanium mesh. These membranes provide excellent structural stability but require a second surgical procedure for removal.
Resorbable membranes are typically composed of collagen or synthetic biodegradable polymers such as polylactide-co-glycolide. These membranes gradually degrade within the body, eliminating the need for a second surgery.
Clinical studies have shown that barrier membranes can significantly improve socket fill and reduce alveolar ridge resorption. However, membrane exposure during healing may compromise treatment outcomes and increase the risk of infection.
3.4 Combination of Bone Graft and Membranes
Combining bone graft materials with barrier membranes is a widely used strategy in ridge preservation. This approach leverages the structural support of graft materials and the cellular exclusion properties of membranes.
Clinical studies have demonstrated that using both materials together can improve bone regeneration and maintain ridge dimensions more effectively than using either approach alone. In such procedures, the graft material fills the socket and provides an osteoconductive scaffold, while the membrane protects the site from soft tissue invasion.
Research has also emphasized the importance of membrane stability. Stabilization techniques, such as fixation with bioabsorbable tacks, have been shown to reduce ridge resorption and improve bone fill by maintaining the membrane in its intended position.
3.5 Tooth Bone Graft
Autogenous tooth-derived grafts have recently emerged as a promising alternative to conventional bone graft materials. Extracted teeth, which were traditionally considered biological waste, contain dentin with chemical and structural characteristics similar to bone.
Dentin consists largely of type I collagen and contains various growth factors, including bone morphogenetic proteins, fibroblast growth factors, and insulin-like growth factors. These components contribute to both osteoconductive and osteoinductive properties.
Tooth-derived graft materials can be processed into several forms, including undemineralized dentin, partially demineralized dentin matrix, and fully demineralized dentin matrix. These materials are biocompatible and do not induce immunological reactions because they are acellular and avascular after processing.
Clinical studies have demonstrated that dentin-derived grafts can effectively support bone regeneration and maintain alveolar ridge dimensions. In some cases, combining dentin matrix with platelet-rich plasma or recombinant growth factors further enhances regenerative potential.
3.6 Autologous Platelet Concentrates (APCs)
Autologous platelet concentrates are biologically active products derived from the patient’s own blood. These materials contain a fibrin network enriched with platelets, leukocytes, and numerous growth factors that regulate tissue healing.
Common forms of platelet concentrates include platelet-rich plasma (PRP), platelet-rich fibrin (PRF), and leukocyte-rich variants of these products. When combined with bone graft materials, they can form a cohesive mixture often referred to as “sticky bone,” which is easier to manipulate during surgical procedures.
APCs release growth factors such as PDGF, TGF-β, and VEGF that promote angiogenesis, cell migration, and tissue regeneration. Clinical studies have shown that platelet concentrates can accelerate soft tissue healing and reduce postoperative discomfort.
However, their effect on preserving alveolar bone volume remains controversial. While some studies report improved bone formation, others have found minimal or inconsistent benefits regarding hard tissue preservation.
3.7 Growth Factors
Growth factors are signaling molecules that regulate key cellular processes such as proliferation, differentiation, migration, and survival. In regenerative dentistry, several growth factors have been investigated for their potential to enhance bone formation.
Among the most extensively studied are bone morphogenetic proteins (BMPs), transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF), and insulin-like growth factor (IGF).
Bone morphogenetic proteins, particularly BMP-2, BMP-4, and BMP-7, have demonstrated strong osteoinductive properties. These proteins can stimulate undifferentiated mesenchymal stem cells to differentiate into osteoblasts, thereby promoting new bone formation.
PDGF, produced by platelets and endothelial cells, primarily stimulates cell proliferation and angiogenesis. Although its osteoinductive capacity is lower than that of BMPs, PDGF plays a critical role in early wound healing and tissue regeneration.
Growth factors are often combined with scaffold materials such as collagen sponges, allografts, or dentin matrices to enhance their effectiveness and control their release within the extraction socket.
3.8 Stem Cells and Tissue Engineering
Tissue engineering represents one of the most advanced approaches to alveolar ridge preservation. This interdisciplinary field integrates principles from biology, medicine, and engineering to develop biological substitutes capable of restoring tissue function.
Tissue engineering strategies generally involve three essential components: stem cells, scaffolds, and signaling molecules. Stem cells provide the cellular basis for regeneration, scaffolds offer structural support, and signaling molecules regulate cell differentiation and tissue formation.
In dentistry, stem cells can be harvested from various oral tissues, including the periodontal ligament, dental pulp, gingiva, and oral epithelium. These cells possess the capacity for self-renewal and can differentiate into multiple cell lineages.
Experimental studies have shown that bone marrow–derived mesenchymal stem cells combined with platelet-rich fibrin can produce injectable bone precursors capable of forming new bone within extraction sockets. Animal studies have also demonstrated accelerated bone healing when stem cells are transplanted into extraction sites.
Although these findings are promising, further clinical research is required to establish the long-term safety and effectiveness of stem cell therapies in routine dental practice.
The future of socket regeneration is likely to involve the integration of stem cell technologies with growth factors and advanced biomaterials, potentially enabling predictable regeneration of alveolar bone with minimal surgical intervention.
Conclusion
Alveolar ridge resorption following tooth extraction remains a significant challenge in implant dentistry. Without intervention, substantial horizontal and vertical bone loss can occur within the first six months after extraction, complicating future restorative procedures.
Modern socket preservation techniques offer clinicians multiple strategies to mitigate these changes. Options range from immediate implant placement and bone grafting to advanced regenerative therapies involving membranes, platelet concentrates, growth factors, and tissue engineering.
Among these methods, bone grafts combined with barrier membranes remain the most widely used and well-documented approach in clinical practice. Emerging technologies, such as dentin-derived grafts, growth factor delivery systems, and stem cell therapies, represent promising advancements that may further enhance regenerative outcomes.
For dental professionals and implantologists, selecting the most appropriate ridge preservation technique requires careful evaluation of patient-specific factors, defect morphology, and long-term treatment goals. Continued research and technological innovation will likely refine these techniques and improve the predictability of alveolar ridge regeneration.
FAQ
1. Why is alveolar ridge preservation important after tooth extraction?
Alveolar ridge preservation helps prevent the significant bone loss that typically occurs following extraction. Maintaining ridge volume simplifies future implant placement and improves esthetic outcomes.
2. When should socket preservation procedures be performed?
Socket preservation is typically performed immediately after tooth extraction, particularly when delayed implant placement is planned or when the risk of ridge resorption is high.
3. What is considered the gold standard bone graft material?
Autogenous bone grafts are widely regarded as the gold standard because they possess osteogenic, osteoinductive, and osteoconductive properties.
4. Do immediate implants prevent bone resorption?
Immediate implants can reduce treatment time but do not completely prevent post-extraction bone remodeling, especially on the buccal plate.
5. Are platelet concentrates effective for socket preservation?
Platelet concentrates can improve soft tissue healing, but evidence regarding their ability to preserve alveolar bone volume remains inconsistent.
6. What is the future of socket regeneration?
Future developments will likely focus on tissue engineering approaches that combine stem cells, biomaterials, and growth factors to achieve predictable bone regeneration.
Reference
Techniques for Extraction Socket Regeneration for Alveolar Ridge Preservation.
Jafer MA, Salem RMA, Hakami FB, Ageeli RE, Alhazmi TA, Bhandi S, Patil S.
Recent Articles
Introduction
The alveolar bone surrounding a tooth is a highly specialized structure characterized by a distinctive microarchitecture. One of its defining features is the dual orientation of collagen fibers created by the insertion of Sharpey’s fibers into the bone matrix. In addition, the alveolar bone is richly vascularized and innervated, with numerous blood vessels and nerve fibers penetrating the structure. Because teeth are constantly subjected to functional loading and biomechanical forces, the surrounding bone undergoes continuous remodeling to maintain structural integrity and adapt to mechanical demands.
The morphology and volume of the alveolar bone are closely influenced by the characteristics of the associated tooth, including its inclination, eruption axis, and anatomical form. When a tooth is extracted, the surrounding alveolar socket inevitably undergoes a cascade of biological and structural changes. These post-extraction events lead to measurable alterations in both the vertical and horizontal dimensions of the alveolar ridge. As a consequence, the available bone volume may be significantly reduced over time.
Such dimensional changes can complicate subsequent implant therapy. Insufficient bone width or height can make implant placement technically challenging, compromise primary stability, and potentially affect osseointegration. Furthermore, loss of alveolar bone volume may negatively influence the esthetic outcome of prosthodontic rehabilitation, whether through implant-supported restorations or conventional prostheses.
Clinical evidence highlights the magnitude of this post-extraction remodeling. A systematic review by Niklaus P. Lang and colleagues quantified the dimensional alterations occurring after tooth removal. Their analysis demonstrated that within six months following extraction, horizontal bone loss typically ranges between approximately 2.5 mm and 4.5 mm, while vertical loss averages around 0.8–1.5 mm. The buccal plate tends to experience more pronounced resorption compared with lingual or palatal surfaces, and buccolingual shrinkage is usually greater than mesiodistal reduction. Importantly, the majority of this bone loss occurs during the first three to six months after extraction, after which the rate of resorption gradually slows.
To counteract these dimensional changes, socket preservation procedures have been developed to maintain the structural integrity of the alveolar ridge. These interventions aim to limit early bone resorption and preserve ridge volume immediately after tooth removal. According to the definition proposed by Daniel S. Thoma Hammerle and colleagues, ridge preservation refers to maintaining the existing ridge dimensions within the envelope present at the time of extraction.
Clinical indications for ridge preservation include cases in which immediate or early implant placement is not feasible, when primary implant stability cannot be achieved, or when prosthodontic treatment requires preservation of ridge contour. With the rapid growth of implant dentistry, ridge preservation is increasingly considered an essential step following extraction in many clinical scenarios. By maintaining both hard and soft tissue architecture, socket preservation simplifies future implant placement and improves functional and esthetic outcomes.
The purpose of this review is to outline the most current extraction socket regeneration techniques and to provide dental professionals and implantologists with an evidence-based overview of modern alveolar ridge preservation strategies.
Healing of the Extraction Socket
A comprehensive understanding of extraction socket healing is fundamental for clinicians seeking to implement effective regeneration or preservation techniques. The healing process follows a well-defined sequence of biological events that involve both resorptive and regenerative mechanisms.
Immediately after tooth extraction, the socket becomes filled with blood, forming a coagulum. This blood clot acts as a temporary scaffold and is subsequently replaced by a provisional matrix composed of fibrin and inflammatory cells. The provisional matrix supports cellular migration and provides the initial framework for tissue regeneration.
During the early stages of healing, osteogenic cells begin to populate the socket, leading to the formation of woven bone. This immature bone tissue gradually bridges the socket and provides the first hard tissue structure within the extraction site. Woven bone is characterized by a disorganized collagen framework and relatively low mechanical strength.
As healing progresses, the remodeling phase begins. During this stage, osteoclastic activity resorbs the immature woven bone while osteoblastic cells deposit more organized lamellar bone. Successive layers of lamellar bone strengthen the newly formed hard tissue bridge and contribute to the maturation of the socket structure.
Eventually, the cortical bone plate begins to develop along the outer boundaries of the socket. Simultaneously, the periosteum forms, and the overlying mucosal tissues attach to the cortical surface. This process, in which bone remodeling results in the development of a cortical wall around the socket, is known as corticalization.
The sequence of clot formation, woven bone deposition, and lamellar bone maturation closely resembles the healing process observed in long-bone fractures. Understanding this physiological progression allows clinicians to design regenerative strategies that enhance osteogenesis while minimizing alveolar ridge resorption.
Socket Preservation Techniques
Multiple techniques and biomaterials have been proposed to preserve the alveolar ridge following tooth extraction. Over the past few decades, advances in biomaterials, molecular biology, and regenerative dentistry have significantly expanded the available therapeutic options.
Socket preservation strategies now range from minimally invasive surgical approaches, such as flapless extraction, to sophisticated regenerative procedures involving biomaterials, biologic mediators, and cell-based therapies. These techniques aim to stabilize the extraction socket, promote bone regeneration, and limit the natural resorptive processes that occur after tooth removal.
Modern alveolar ridge preservation approaches include immediate implant placement, bone grafting, barrier membrane application, platelet-derived biologics, growth factor therapies, tooth-derived graft materials, and emerging tissue engineering techniques involving stem cells.
3.1 Immediate Implants as Socket Preservers
Traditionally, dental implant placement is delayed for approximately six to twelve months following tooth extraction to allow complete healing of the socket. However, during this healing period, substantial resorption of the alveolar ridge may occur, potentially complicating later implant placement and compromising treatment outcomes.
Immediate implant placement has been proposed as a strategy to reduce post-extraction bone resorption by occupying the socket immediately after extraction. The rationale is that the implant fixture may help maintain alveolar ridge dimensions and minimize bone remodeling.
Some clinicians have advocated immediate implant placement without additional preservation procedures such as barrier membranes or graft materials. This approach was suggested as a means to prevent dimensional changes in the alveolar ridge while simultaneously shortening treatment time.
However, clinical and experimental studies have reported mixed outcomes regarding the effectiveness of immediate implants in preventing bone loss. Research conducted by Marcelo Araujo demonstrated that even when implants were placed immediately after extraction, measurable bone loss still occurred during the healing phase. In experimental models, approximately 2.6 mm of bone reduction was observed after three months of healing.
Additional investigations showed that while initial bone-to-implant contact could be established within one month, resorption of the buccal plate often occurred subsequently. As the buccal bone wall resorbed, some of the previously established bone-to-implant contact was partially lost during later stages of healing.
Clinical research conducted by Tiziano Botticelli further confirmed that immediate implant placement does not completely prevent the physiological bone remodeling that occurs after extraction. Even when implants were immediately loaded, the extent of buccal bone resorption remained largely unchanged.
These findings indicate that although immediate implants offer advantages such as reduced treatment time and fewer surgical interventions, they do not fully eliminate post-extraction bone resorption. As a result, clinicians often combine immediate implantation with additional ridge preservation techniques to improve outcomes.
3.2 Bone Grafts and Bone Substitutes
Bone grafting remains one of the most widely used approaches for extraction socket regeneration. These materials function primarily as scaffolds that support new bone formation while limiting the collapse of the socket walls.
Autogenous Bone Grafts
Autogenous bone grafts are harvested directly from the patient and are widely regarded as the gold standard in bone regeneration. Common intraoral donor sites include the maxillary tuberosity, mandibular ramus, mandibular symphysis, and edentulous ridges. Extraoral donor sites such as the iliac crest or tibia may also be used when larger volumes are required.
Autogenous grafts possess osteoconductive, osteoinductive, and osteogenic properties. They support bone formation by providing living cells, growth factors, and a structural scaffold. Additionally, they are biocompatible and do not provoke immunological reactions.
Allogenic Bone Grafts
Allogenic grafts, or allografts, are obtained from donors of the same species. These grafts are typically processed through techniques such as freeze-drying or demineralization to ensure safety and reduce antigenicity.
Demineralized freeze-dried bone allograft (DFDBA) is particularly notable for its osteoinductive potential because the demineralization process exposes bone morphogenetic proteins. However, clinicians must consider the theoretical risk of disease transmission and immune response associated with donor-derived materials.
Xenografts and Alloplastic Materials
Bone substitutes also include xenografts and alloplastic materials. Xenografts originate from different species, commonly bovine, porcine, or equine, and function primarily as osteoconductive scaffolds.
Alloplastic materials are synthetic bone substitutes manufactured under controlled laboratory conditions. These materials include bioceramics such as hydroxyapatite and biphasic calcium phosphate, as well as biodegradable polymers. Because they are synthetically produced, their structural properties, including porosity and degradation rate, can be precisely controlled.
Numerous studies have demonstrated that these materials can effectively reduce alveolar ridge resorption when used as socket fillers, although the degree of new bone formation varies depending on the biomaterial used.
3.3 Barrier Membranes in Socket Preservation
The concept of barrier membranes in regenerative dentistry originates from the principle that different cell populations compete to colonize healing wounds. According to a hypothesis proposed by Melvin Melcher, the migration of non-osteogenic cells, such as gingival epithelial cells, into a bone defect may inhibit bone regeneration by preventing osteogenic cells from populating the site.
To address this issue, barrier membranes were introduced to selectively exclude undesirable cell types and create a protected environment for bone regeneration. This concept led to the development of guided tissue regeneration (GTR), later adapted into guided bone regeneration (GBR) for implant dentistry.
In GBR procedures, a membrane is placed over the extraction socket to isolate the defect and maintain space for osteoprogenitor cells. By preventing soft tissue invasion, the membrane allows osteogenic cells to proliferate and differentiate, thereby enhancing new bone formation.
Barrier membranes are broadly categorized into non-resorbable and resorbable types.
Non-resorbable membranes include materials such as expanded polytetrafluoroethylene (ePTFE) and titanium mesh. These membranes provide excellent structural stability but require a second surgical procedure for removal.
Resorbable membranes are typically composed of collagen or synthetic biodegradable polymers such as polylactide-co-glycolide. These membranes gradually degrade within the body, eliminating the need for a second surgery.
Clinical studies have shown that barrier membranes can significantly improve socket fill and reduce alveolar ridge resorption. However, membrane exposure during healing may compromise treatment outcomes and increase the risk of infection.
3.4 Combination of Bone Graft and Membranes
Combining bone graft materials with barrier membranes is a widely used strategy in ridge preservation. This approach leverages the structural support of graft materials and the cellular exclusion properties of membranes.
Clinical studies have demonstrated that using both materials together can improve bone regeneration and maintain ridge dimensions more effectively than using either approach alone. In such procedures, the graft material fills the socket and provides an osteoconductive scaffold, while the membrane protects the site from soft tissue invasion.
Research has also emphasized the importance of membrane stability. Stabilization techniques, such as fixation with bioabsorbable tacks, have been shown to reduce ridge resorption and improve bone fill by maintaining the membrane in its intended position.
3.5 Tooth Bone Graft
Autogenous tooth-derived grafts have recently emerged as a promising alternative to conventional bone graft materials. Extracted teeth, which were traditionally considered biological waste, contain dentin with chemical and structural characteristics similar to bone.
Dentin consists largely of type I collagen and contains various growth factors, including bone morphogenetic proteins, fibroblast growth factors, and insulin-like growth factors. These components contribute to both osteoconductive and osteoinductive properties.
Tooth-derived graft materials can be processed into several forms, including undemineralized dentin, partially demineralized dentin matrix, and fully demineralized dentin matrix. These materials are biocompatible and do not induce immunological reactions because they are acellular and avascular after processing.
Clinical studies have demonstrated that dentin-derived grafts can effectively support bone regeneration and maintain alveolar ridge dimensions. In some cases, combining dentin matrix with platelet-rich plasma or recombinant growth factors further enhances regenerative potential.
3.6 Autologous Platelet Concentrates (APCs)
Autologous platelet concentrates are biologically active products derived from the patient’s own blood. These materials contain a fibrin network enriched with platelets, leukocytes, and numerous growth factors that regulate tissue healing.
Common forms of platelet concentrates include platelet-rich plasma (PRP), platelet-rich fibrin (PRF), and leukocyte-rich variants of these products. When combined with bone graft materials, they can form a cohesive mixture often referred to as “sticky bone,” which is easier to manipulate during surgical procedures.
APCs release growth factors such as PDGF, TGF-β, and VEGF that promote angiogenesis, cell migration, and tissue regeneration. Clinical studies have shown that platelet concentrates can accelerate soft tissue healing and reduce postoperative discomfort.
However, their effect on preserving alveolar bone volume remains controversial. While some studies report improved bone formation, others have found minimal or inconsistent benefits regarding hard tissue preservation.
3.7 Growth Factors
Growth factors are signaling molecules that regulate key cellular processes such as proliferation, differentiation, migration, and survival. In regenerative dentistry, several growth factors have been investigated for their potential to enhance bone formation.
Among the most extensively studied are bone morphogenetic proteins (BMPs), transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF), and insulin-like growth factor (IGF).
Bone morphogenetic proteins, particularly BMP-2, BMP-4, and BMP-7, have demonstrated strong osteoinductive properties. These proteins can stimulate undifferentiated mesenchymal stem cells to differentiate into osteoblasts, thereby promoting new bone formation.
PDGF, produced by platelets and endothelial cells, primarily stimulates cell proliferation and angiogenesis. Although its osteoinductive capacity is lower than that of BMPs, PDGF plays a critical role in early wound healing and tissue regeneration.
Growth factors are often combined with scaffold materials such as collagen sponges, allografts, or dentin matrices to enhance their effectiveness and control their release within the extraction socket.
3.8 Stem Cells and Tissue Engineering
Tissue engineering represents one of the most advanced approaches to alveolar ridge preservation. This interdisciplinary field integrates principles from biology, medicine, and engineering to develop biological substitutes capable of restoring tissue function.
Tissue engineering strategies generally involve three essential components: stem cells, scaffolds, and signaling molecules. Stem cells provide the cellular basis for regeneration, scaffolds offer structural support, and signaling molecules regulate cell differentiation and tissue formation.
In dentistry, stem cells can be harvested from various oral tissues, including the periodontal ligament, dental pulp, gingiva, and oral epithelium. These cells possess the capacity for self-renewal and can differentiate into multiple cell lineages.
Experimental studies have shown that bone marrow–derived mesenchymal stem cells combined with platelet-rich fibrin can produce injectable bone precursors capable of forming new bone within extraction sockets. Animal studies have also demonstrated accelerated bone healing when stem cells are transplanted into extraction sites.
Although these findings are promising, further clinical research is required to establish the long-term safety and effectiveness of stem cell therapies in routine dental practice.
The future of socket regeneration is likely to involve the integration of stem cell technologies with growth factors and advanced biomaterials, potentially enabling predictable regeneration of alveolar bone with minimal surgical intervention.
Conclusion
Alveolar ridge resorption following tooth extraction remains a significant challenge in implant dentistry. Without intervention, substantial horizontal and vertical bone loss can occur within the first six months after extraction, complicating future restorative procedures.
Modern socket preservation techniques offer clinicians multiple strategies to mitigate these changes. Options range from immediate implant placement and bone grafting to advanced regenerative therapies involving membranes, platelet concentrates, growth factors, and tissue engineering.
Among these methods, bone grafts combined with barrier membranes remain the most widely used and well-documented approach in clinical practice. Emerging technologies, such as dentin-derived grafts, growth factor delivery systems, and stem cell therapies, represent promising advancements that may further enhance regenerative outcomes.
For dental professionals and implantologists, selecting the most appropriate ridge preservation technique requires careful evaluation of patient-specific factors, defect morphology, and long-term treatment goals. Continued research and technological innovation will likely refine these techniques and improve the predictability of alveolar ridge regeneration.
FAQ
1. Why is alveolar ridge preservation important after tooth extraction?
Alveolar ridge preservation helps prevent the significant bone loss that typically occurs following extraction. Maintaining ridge volume simplifies future implant placement and improves esthetic outcomes.
2. When should socket preservation procedures be performed?
Socket preservation is typically performed immediately after tooth extraction, particularly when delayed implant placement is planned or when the risk of ridge resorption is high.
3. What is considered the gold standard bone graft material?
Autogenous bone grafts are widely regarded as the gold standard because they possess osteogenic, osteoinductive, and osteoconductive properties.
4. Do immediate implants prevent bone resorption?
Immediate implants can reduce treatment time but do not completely prevent post-extraction bone remodeling, especially on the buccal plate.
5. Are platelet concentrates effective for socket preservation?
Platelet concentrates can improve soft tissue healing, but evidence regarding their ability to preserve alveolar bone volume remains inconsistent.
6. What is the future of socket regeneration?
Future developments will likely focus on tissue engineering approaches that combine stem cells, biomaterials, and growth factors to achieve predictable bone regeneration.
Reference
Techniques for Extraction Socket Regeneration for Alveolar Ridge Preservation.
Jafer MA, Salem RMA, Hakami FB, Ageeli RE, Alhazmi TA, Bhandi S, Patil S.
Recent Articles
Pulsed Electromagnetic Field (PEMF) Use in Implant Dentistry
Pulsed Electromagnetic Field (PEMF) Use in Implant Dentistry Abstract and Historical Background Pulsed Electromagnetic Field (PEMF) therapy refers to the application of low-frequency electromagnetic [...]
Laser Therapy in the Management of Peri-Implantitis
Laser Therapy in the Management of Peri-Implantitis Laser technology has become an increasingly valuable adjunct in the treatment of peri-implant diseases, particularly peri-implantitis. Its [...]
Magnetically Guided Dental Nanobots (CalBots) Are Real
Magnetically Guided Dental Nanobots (CalBots) Are Real The concept of nanobots in dentistry has long been associated with futuristic speculation. However, recent peer-reviewed research [...]
Peri-Implantitis Treatment & Prevention Methods
Peri-Implantitis Treatment & Prevention Methods Peri-implantitis remains one of the most significant biological complications affecting dental implants, posing a serious threat to long-term implant [...]
Latest Sinus Lifting Techniques in Modern Implant Dentistry
Latest Sinus Lifting Techniques in Modern Implant Dentistry A Comprehensive Clinical Review Introduction to Sinus Lifting in Implantology Sinus lifting, or maxillary sinus floor [...]
Biomechanics of the Triple Abutment & BOPiT Concept
Dental Biomechanics · Implant Science · Clinical Evidence Biomechanics of the Triple Abutment & BOPiT Concept How a saddle-shaped mathematical surface is rewriting the rules of load distribution [...]