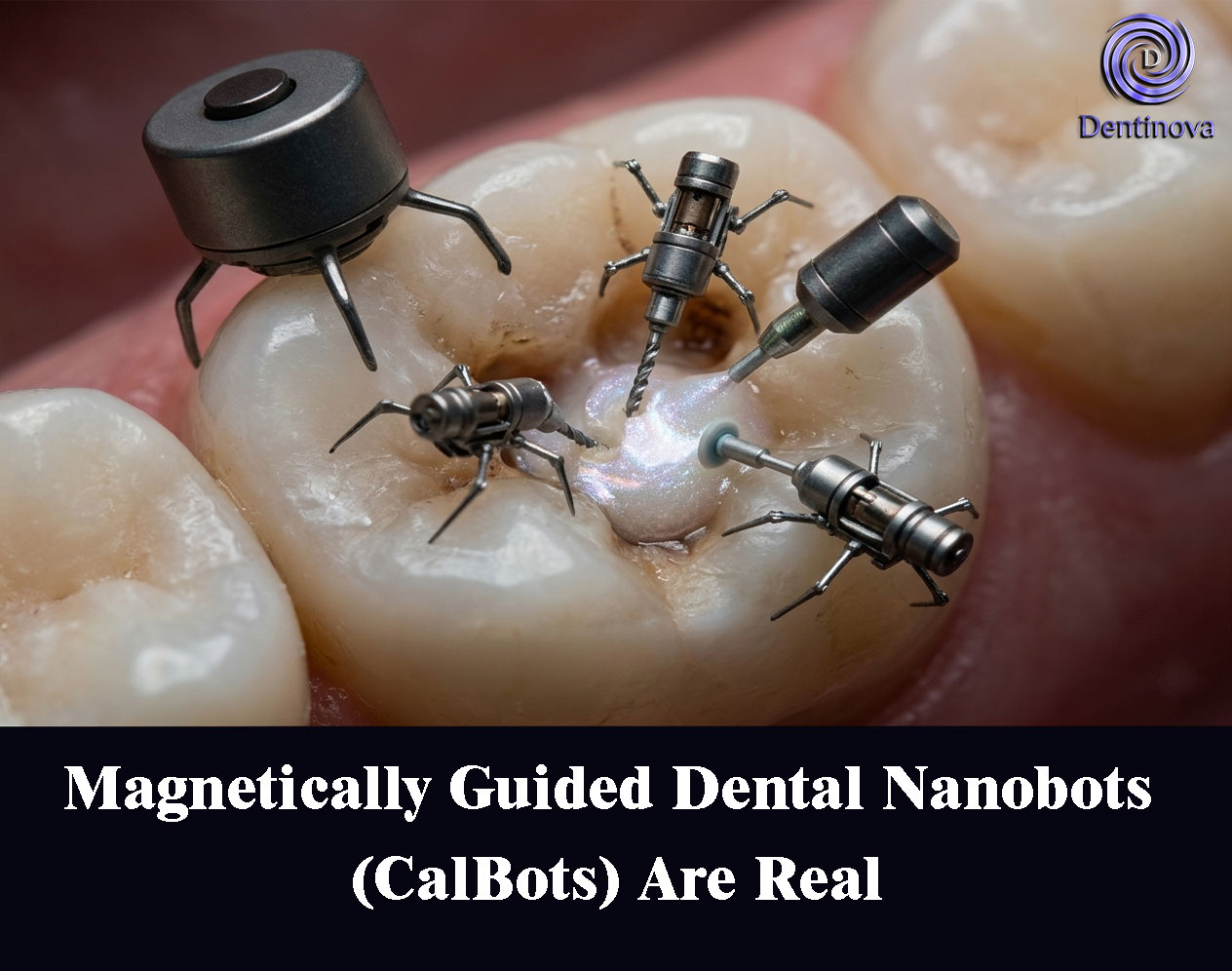
Magnetically Guided Dental Nanobots (CalBots) Are Real
The concept of nanobots in dentistry has long been associated with futuristic speculation. However, recent peer-reviewed research published in the journal Advanced Science (2025) has brought this vision closer to clinical reality. A groundbreaking study titled “Directed Self-Assembly of Magnetic Bioceramic Deep Inside Dentinal Tubules May Alleviate Dental Hypersensitivity” demonstrates that magnetically guided nanomaterials can actively penetrate dentinal tubules and form durable, biocompatible seals. This innovation represents a paradigm shift in the management of dentinal hypersensitivity, moving from superficial symptom control to targeted, deep-tissue intervention.
Understanding Dentinal Hypersensitivity: A Global Clinical Challenge
Dentinal hypersensitivity (DH) is a highly prevalent condition affecting over one billion individuals worldwide. It is characterized by sharp, transient pain triggered by thermal, chemical, or tactile stimuli such as cold air, acidic beverages, or tooth brushing. The underlying mechanism is well explained by the hydrodynamic theory, where fluid movement within exposed dentinal tubules stimulates nerve endings located in the pulp.
This exposure typically results from enamel erosion, gingival recession, or loss of cementum. Once dentinal tubules are open to the oral environment, they act as direct pathways to the pulp, amplifying sensitivity and discomfort. Despite its widespread occurrence, DH remains challenging to treat effectively with long-term success.
Limitations of Conventional Treatment Modalities
Current approaches to managing dentinal hypersensitivity primarily focus on occluding dentinal tubules. These include desensitizing toothpastes, adhesive resins, and laser treatments. However, these methods share significant limitations:
- They provide only superficial tubule occlusion
- Require continuous reapplication
- Show limited long-term efficacy
- Fail to penetrate deeply into the dentinal structure
For example, resin-based sealers demonstrate less than 10% effectiveness after four weeks, while desensitizing toothpastes require consistent use over several weeks to achieve minimal relief. Laser treatments, although innovative, may cause thermal damage to dentin and do not reliably achieve durable sealing.
The fundamental limitation lies in the inability of these treatments to navigate the complex microstructure of dentin, which consists of densely packed tubules with varying orientations and diameters. This challenge has historically prevented deep, stable occlusion—until now.
Introducing CalBots: Magnetically Guided Dental Nanobots
The recent study introduces a novel nanomaterial platform known as CalBots (Calcium-based nanobots). These are superparamagnetic, bioceramic nanoparticles engineered to achieve controlled navigation and self-assembly within dentinal tubules.
Each CalBot consists of:
- An iron oxide core enabling magnetic responsiveness
- A silica shell providing structural stability
- An outer layer of calcium silicate, essential for bioceramic sealing
With an average diameter of approximately 387 nm, these particles are small enough to enter dentinal tubules yet capable of forming larger functional structures through magnetic manipulation.
How Magnetic Guidance Enables Deep Tubule Penetration
The key innovation lies in the ability to actively control the movement of CalBots using an external magnetic field. When exposed to a magnetic gradient, these nanoparticles undergo directed self-assembly, forming chain-like structures that can be propelled deep into dentinal tubules.
Unlike passive diffusion-based systems, this active approach allows penetration depths exceeding 300–500 micrometers, significantly surpassing traditional treatments. The process is highly dependent on optimizing parameters such as:
- Magnetic field strength
- Particle concentration
- Chain length dynamics
Proper tuning ensures that CalBots form flexible chains rather than bulky aggregates, enabling them to navigate the پیچیدہ topography of dentin effectively.
Bioceramic Plug Formation: A Regenerative Mechanism
Once inside the dentinal tubules, CalBots initiate a chemical transformation that leads to the formation of a calcium silicate hydrate gel. This process occurs in the presence of calcium oxide and ambient conditions, resulting in a self-setting, cement-like structure.
This bioceramic matrix:
- Forms a robust, stable plug within the tubule
- Is biocompatible and similar to materials used in regenerative dentistry
- Provides long-lasting occlusion resistant to external factors
Importantly, this mechanism goes beyond simple coating. It represents true intratubular regeneration, effectively sealing the pathway responsible for sensitivity at its source.
Quantitative Results: Significant Reduction in Dentin Permeability
Experimental data from ex vivo human teeth demonstrated remarkable outcomes:
- Up to 87.7% reduction in dentin permeability
- Occlusion depths reaching 500 µm
- Up to 90% reduction in open tubules at significant depths
Advanced imaging techniques, including scanning electron microscopy (SEM) and micro-CT analysis, confirmed the formation of dense, well-distributed plugs throughout the dentinal structure.
In Vivo Evidence: Complete Functional Recovery in Animal Models
The study also included controlled animal trials using a murine model to evaluate clinical relevance. Dentinal hypersensitivity was induced in mice, followed by treatment with magnetically guided CalBots.
The results were striking:
- Full behavioral recovery from hypersensitivity
- Restoration of normal response to cold stimuli
- Significant improvement across multiple behavioral metrics
Unlike conventional treatments, which showed only temporary or minimal effects, CalBot therapy provided sustained relief, with benefits observed for at least two weeks post-treatment.
Safety Profile and Biocompatibility
Safety is a critical factor in any emerging dental technology. The study conducted comprehensive toxicity assessments using standardized protocols. Findings revealed:
- No mortality or adverse effects in the animal models
- Normal blood parameters and histological findings
- High biocompatibility due to use of GRAS materials
These results suggest strong potential for safe translation into human clinical applications, although further trials are required.
Clinical Implications: A New Era in Minimally Invasive Dentistry
If successfully translated into clinical practice, magnetically guided dental nanobots could redefine the standard of care for dentinal hypersensitivity. Potential advantages include:
- Non-invasive treatment without drilling
- Rapid procedure time (10–20 minutes)
- Long-lasting results due to deep sealing
- Targeted therapy addressing root cause
A typical clinical workflow may involve applying a CalBot suspension to exposed dentin, followed by activation using a handheld magnetic device to guide particles into tubules.
Beyond Hypersensitivity: Broader Applications in Dentistry
The implications of this technology extend beyond sensitivity management. The formation of deep, impermeable plugs may:
- Prevent bacterial infiltration into dentin
- Enhance protection of the pulp
- Support early intervention in dentinal caries
- Provide benefits in conditions like amelogenesis imperfecta
This positions CalBots as a potential platform technology for regenerative and preventive dentistry.
Challenges and Future Directions
Despite its promise, this technology is still in the pre-clinical stage. Several challenges must be addressed before widespread adoption:
- Human clinical trials to confirm efficacy and safety
- Optimization of magnetic delivery systems
- Regulatory approval and standardization
- Long-term durability studies under oral conditions
Future research will also explore integration with other regenerative therapies and expansion into additional medical applications involving calcified tissues.
Conclusion: From Science Fiction to Clinical Reality
Magnetically guided dental nanobots are no longer theoretical constructs—they are experimentally validated tools with the potential to transform modern dentistry. By combining nanotechnology, magnetic control, and bioceramic chemistry, this approach addresses one of the most persistent challenges in dental care with unprecedented precision.
While further research is required before clinical implementation, the evidence clearly indicates that we are entering a new era where microscopic, intelligent materials can perform targeted treatment deep inside tooth structure. The future of dentistry is not just minimally invasive—it is microscopically precise.
FAQ: Magnetically Guided Dental Nanobots (CalBots)
1. What are magnetically guided dental nanobots?
Magnetically guided dental nanobots, known as CalBots, are superparamagnetic calcium-based bioceramic nanoparticles designed to enter dentinal tubules and be controlled using an external magnetic field. They can self-assemble into chains and penetrate deep into tooth structure to treat dentinal hypersensitivity at its source.
2. How do CalBots treat dental hypersensitivity?
CalBots work by entering exposed dentinal tubules and forming a bioceramic calcium silicate hydrate plug. This plug blocks fluid movement inside the tubules, which is the main cause of pain in dentinal hypersensitivity, effectively reducing nerve stimulation and eliminating sensitivity triggers.
3. How deep can CalBots penetrate into dentinal tubules?
Experimental results show that CalBots can penetrate more than 300–500 micrometers into dentinal tubules under a controlled magnetic field. This depth is significantly greater than conventional treatments, which typically act only on the surface of the tooth.
4. Are CalBots more effective than traditional treatments?
Yes, according to the study, CalBots demonstrate significantly higher and more durable effectiveness compared to toothpaste, resin sealers, and laser treatments. They achieved up to 87.7% reduction in dentin permeability and showed sustained relief in animal models.
5. Are magnetically guided nanobots safe for clinical use?
Preclinical animal studies showed that CalBots are biocompatible and did not cause toxicity, behavioral distress, or abnormal blood parameters at tested doses. However, they are still in the research phase and require further clinical trials before being used in human patients.
Reference:
Recent Articles
The concept of nanobots in dentistry has long been associated with futuristic speculation. However, recent peer-reviewed research published in the journal Advanced Science (2025) has brought this vision closer to clinical reality. A groundbreaking study titled “Directed Self-Assembly of Magnetic Bioceramic Deep Inside Dentinal Tubules May Alleviate Dental Hypersensitivity” demonstrates that magnetically guided nanomaterials can actively penetrate dentinal tubules and form durable, biocompatible seals. This innovation represents a paradigm shift in the management of dentinal hypersensitivity, moving from superficial symptom control to targeted, deep-tissue intervention.
Understanding Dentinal Hypersensitivity: A Global Clinical Challenge
Dentinal hypersensitivity (DH) is a highly prevalent condition affecting over one billion individuals worldwide. It is characterized by sharp, transient pain triggered by thermal, chemical, or tactile stimuli such as cold air, acidic beverages, or tooth brushing. The underlying mechanism is well explained by the hydrodynamic theory, where fluid movement within exposed dentinal tubules stimulates nerve endings located in the pulp.
This exposure typically results from enamel erosion, gingival recession, or loss of cementum. Once dentinal tubules are open to the oral environment, they act as direct pathways to the pulp, amplifying sensitivity and discomfort. Despite its widespread occurrence, DH remains challenging to treat effectively with long-term success.
Limitations of Conventional Treatment Modalities
Current approaches to managing dentinal hypersensitivity primarily focus on occluding dentinal tubules. These include desensitizing toothpastes, adhesive resins, and laser treatments. However, these methods share significant limitations:
- They provide only superficial tubule occlusion
- Require continuous reapplication
- Show limited long-term efficacy
- Fail to penetrate deeply into the dentinal structure
For example, resin-based sealers demonstrate less than 10% effectiveness after four weeks, while desensitizing toothpastes require consistent use over several weeks to achieve minimal relief. Laser treatments, although innovative, may cause thermal damage to dentin and do not reliably achieve durable sealing.
The fundamental limitation lies in the inability of these treatments to navigate the complex microstructure of dentin, which consists of densely packed tubules with varying orientations and diameters. This challenge has historically prevented deep, stable occlusion—until now.
Introducing CalBots: Magnetically Guided Dental Nanobots
The recent study introduces a novel nanomaterial platform known as CalBots (Calcium-based nanobots). These are superparamagnetic, bioceramic nanoparticles engineered to achieve controlled navigation and self-assembly within dentinal tubules.
Each CalBot consists of:
- An iron oxide core enabling magnetic responsiveness
- A silica shell providing structural stability
- An outer layer of calcium silicate, essential for bioceramic sealing
With an average diameter of approximately 387 nm, these particles are small enough to enter dentinal tubules yet capable of forming larger functional structures through magnetic manipulation.
How Magnetic Guidance Enables Deep Tubule Penetration
The key innovation lies in the ability to actively control the movement of CalBots using an external magnetic field. When exposed to a magnetic gradient, these nanoparticles undergo directed self-assembly, forming chain-like structures that can be propelled deep into dentinal tubules.
Unlike passive diffusion-based systems, this active approach allows penetration depths exceeding 300–500 micrometers, significantly surpassing traditional treatments. The process is highly dependent on optimizing parameters such as:
- Magnetic field strength
- Particle concentration
- Chain length dynamics
Proper tuning ensures that CalBots form flexible chains rather than bulky aggregates, enabling them to navigate the پیچیدہ topography of dentin effectively.
Bioceramic Plug Formation: A Regenerative Mechanism
Once inside the dentinal tubules, CalBots initiate a chemical transformation that leads to the formation of a calcium silicate hydrate gel. This process occurs in the presence of calcium oxide and ambient conditions, resulting in a self-setting, cement-like structure.
This bioceramic matrix:
- Forms a robust, stable plug within the tubule
- Is biocompatible and similar to materials used in regenerative dentistry
- Provides long-lasting occlusion resistant to external factors
Importantly, this mechanism goes beyond simple coating. It represents true intratubular regeneration, effectively sealing the pathway responsible for sensitivity at its source.
Quantitative Results: Significant Reduction in Dentin Permeability
Experimental data from ex vivo human teeth demonstrated remarkable outcomes:
- Up to 87.7% reduction in dentin permeability
- Occlusion depths reaching 500 µm
- Up to 90% reduction in open tubules at significant depths
Advanced imaging techniques, including scanning electron microscopy (SEM) and micro-CT analysis, confirmed the formation of dense, well-distributed plugs throughout the dentinal structure.
In Vivo Evidence: Complete Functional Recovery in Animal Models
The study also included controlled animal trials using a murine model to evaluate clinical relevance. Dentinal hypersensitivity was induced in mice, followed by treatment with magnetically guided CalBots.
The results were striking:
- Full behavioral recovery from hypersensitivity
- Restoration of normal response to cold stimuli
- Significant improvement across multiple behavioral metrics
Unlike conventional treatments, which showed only temporary or minimal effects, CalBot therapy provided sustained relief, with benefits observed for at least two weeks post-treatment.
Safety Profile and Biocompatibility
Safety is a critical factor in any emerging dental technology. The study conducted comprehensive toxicity assessments using standardized protocols. Findings revealed:
- No mortality or adverse effects in the animal models
- Normal blood parameters and histological findings
- High biocompatibility due to use of GRAS materials
These results suggest strong potential for safe translation into human clinical applications, although further trials are required.
Clinical Implications: A New Era in Minimally Invasive Dentistry
If successfully translated into clinical practice, magnetically guided dental nanobots could redefine the standard of care for dentinal hypersensitivity. Potential advantages include:
- Non-invasive treatment without drilling
- Rapid procedure time (10–20 minutes)
- Long-lasting results due to deep sealing
- Targeted therapy addressing root cause
A typical clinical workflow may involve applying a CalBot suspension to exposed dentin, followed by activation using a handheld magnetic device to guide particles into tubules.
Beyond Hypersensitivity: Broader Applications in Dentistry
The implications of this technology extend beyond sensitivity management. The formation of deep, impermeable plugs may:
- Prevent bacterial infiltration into dentin
- Enhance protection of the pulp
- Support early intervention in dentinal caries
- Provide benefits in conditions like amelogenesis imperfecta
This positions CalBots as a potential platform technology for regenerative and preventive dentistry.
Challenges and Future Directions
Despite its promise, this technology is still in the pre-clinical stage. Several challenges must be addressed before widespread adoption:
- Human clinical trials to confirm efficacy and safety
- Optimization of magnetic delivery systems
- Regulatory approval and standardization
- Long-term durability studies under oral conditions
Future research will also explore integration with other regenerative therapies and expansion into additional medical applications involving calcified tissues.
Conclusion: From Science Fiction to Clinical Reality
Magnetically guided dental nanobots are no longer theoretical constructs—they are experimentally validated tools with the potential to transform modern dentistry. By combining nanotechnology, magnetic control, and bioceramic chemistry, this approach addresses one of the most persistent challenges in dental care with unprecedented precision.
While further research is required before clinical implementation, the evidence clearly indicates that we are entering a new era where microscopic, intelligent materials can perform targeted treatment deep inside tooth structure. The future of dentistry is not just minimally invasive—it is microscopically precise.
FAQ: Magnetically Guided Dental Nanobots (CalBots)
1. What are magnetically guided dental nanobots?
Magnetically guided dental nanobots, known as CalBots, are superparamagnetic calcium-based bioceramic nanoparticles designed to enter dentinal tubules and be controlled using an external magnetic field. They can self-assemble into chains and penetrate deep into tooth structure to treat dentinal hypersensitivity at its source.
2. How do CalBots treat dental hypersensitivity?
CalBots work by entering exposed dentinal tubules and forming a bioceramic calcium silicate hydrate plug. This plug blocks fluid movement inside the tubules, which is the main cause of pain in dentinal hypersensitivity, effectively reducing nerve stimulation and eliminating sensitivity triggers.
3. How deep can CalBots penetrate into dentinal tubules?
Experimental results show that CalBots can penetrate more than 300–500 micrometers into dentinal tubules under a controlled magnetic field. This depth is significantly greater than conventional treatments, which typically act only on the surface of the tooth.
4. Are CalBots more effective than traditional treatments?
Yes, according to the study, CalBots demonstrate significantly higher and more durable effectiveness compared to toothpaste, resin sealers, and laser treatments. They achieved up to 87.7% reduction in dentin permeability and showed sustained relief in animal models.
5. Are magnetically guided nanobots safe for clinical use?
Preclinical animal studies showed that CalBots are biocompatible and did not cause toxicity, behavioral distress, or abnormal blood parameters at tested doses. However, they are still in the research phase and require further clinical trials before being used in human patients.
Reference:
Recent Articles
Peri-Implantitis Treatment & Prevention Methods
Peri-Implantitis Treatment & Prevention Methods Peri-implantitis remains one of the most significant biological complications affecting dental implants, posing a serious threat to long-term implant [...]
Latest Sinus Lifting Techniques in Modern Implant Dentistry
Latest Sinus Lifting Techniques in Modern Implant Dentistry A Comprehensive Clinical Review Introduction to Sinus Lifting in Implantology Sinus lifting, or maxillary sinus floor [...]
Biomechanics of the Triple Abutment & BOPiT Concept
Dental Biomechanics · Implant Science · Clinical Evidence Biomechanics of the Triple Abutment & BOPiT Concept How a saddle-shaped mathematical surface is rewriting the rules of load distribution [...]
The Woman Who Proved One Implant Could Hold Three Crowns
The Woman Who Proved One Implant Could Hold Three Crowns While the dental establishment looked away, Dr. Luciana Colepícolo spent 12 years building the [...]
ENPP1, The Molecular Brake That Limits Tissue Healing
ENPP1, The Molecular Brake That Limits Tissue Healing Introduction: A New Biological Barrier to Dental Regeneration Meet ENPP1—a protein most dentists have never heard [...]
The Rise of Self‑Healing Dental Biomaterials
The Rise of Self‑Healing Dental Biomaterials Introduction In modern restorative dentistry, durability and longevity of materials remain among the greatest clinical challenges. Traditional dental [...]