Exploring the Use of Dead Skin in Developing New Gingiva
A Cutting-Edge Perspective for Dental Specialists
As periodontal and gingival surgery expands into regenerative frontiers, the concept of incorporating materials traditionally reserved for dermatologic or tissue-engineering purposes is gaining traction. One of these materials is the use of dead skin or skin allograft tissue in the development of new gingiva, specifically in augmenting keratinized attached gingiva. This article is tailored for dental specialists and explores how this innovative approach can bolster periodontal outcomes, the biological rationale, clinical evidence, and practical considerations. Key search phrase: use of dead skin in developing new gingiva.
1. Understanding the Biological Basis for the use of dead skin in developing new gingiva
To grasp why the use of dead skin in developing new gingiva might be feasible, one must review the histology of both skin and gingiva. The keratinized gingiva acts as a robust barrier that sustains mechanical insult, bacterial challenge, and shear forces. Skin tissue, similarly keratinized, offers a structural paradigm that can be adapted. In tissue‐engineering studies, skin allografts (for example, freeze-dried skin) have been successfully used to treat mucogingival problems, showing increases in the width of keratinized tissue. By applying the principle of “dead skin” (i.e., processed skin allograft) as a scaffold or grafting medium, clinicians may harness the structural and biological benefits of the skin dermal/epidermal complex to regenerate gingival tissue.
2. Clinical Evidence Supporting the use of dead skin in developing new gingiva
Dental specialists looking at the use of dead skin in developing new gingiva should evaluate the available clinical data. One landmark study describes the use of freeze-dried skin (FDS) allograft in human patients with mucogingival defects: 14 patients received 24 FDS allografts over 56 tooth sites, showing a mean gain of keratinized gingiva of approximately 2.88 ± 1.57 mm at 12 months. These results support that skin allografts may serve as a stable alternative for gingival augmentation. Additionally, comparative research of skin versus gingival keratinocyte healing shows that gingival keratinocytes exhibit faster re-epithelialization. Such data suggest that while gingiva may innately regenerate well, the introduction of a skin-derived scaffold may enhance or accelerate that process, giving credence to the concept of the use of dead skin in developing new gingiva.
3. Mechanisms of Action: When You Apply the use of dead skin in developing new gingiva
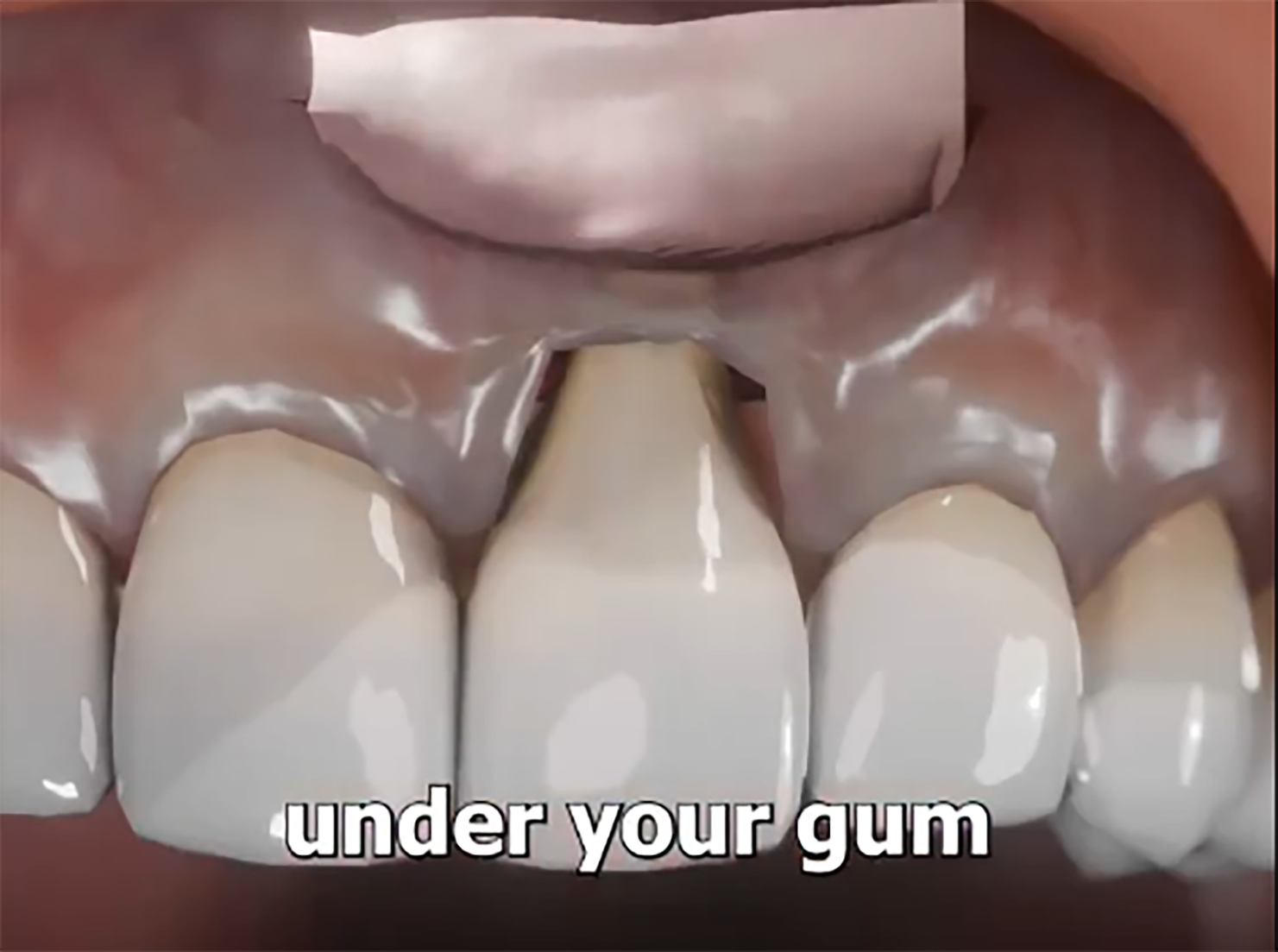
The term “use of dead skin in developing new gingiva” refers to the grafting of devitalized skin tissue (epidermal + dermal layers) onto a prepared gingival bed. The graft acts as a scaffold: the dermal matrix supports vascular ingrowth and fibroblast migration; the epidermal layer may provide a keratinized barrier. Over time, host gingival tissues integrate, remodel, and differentiate, establishing a new zone of keratinized attached gingiva. The superior wound-healing properties of gingiva (versus skin) are well-documented. By placing a “dead skin” graft, you harness both the mechanical structure of skin tissue and the regenerative milieu of the gingiva. Importantly, the scaffold is devitalized (hence “dead”), reducing immune reaction while enabling host cell infiltration and remodeling. The result: a predictable band of keratinized gingiva, improved soft-tissue resilience, and enhanced periodontal stability.
4. Indications and Clinical Protocols for the use of dead skin in developing new gingiva
Dental specialists should consider the use of dead skin in developing new gingiva when facing cases such as inadequate keratinized tissue width, recession with minimal attached gingiva, implant sites lacking soft-tissue bulk, or mucogingival defects resistant to conventional grafting. The clinical protocol may involve: (1) patient assessment and consent regarding allograft use; (2) recipient site preparation (partial‐thickness flap, apical periosteal separation); (3) rehydration and placement of the skin allograft; (4) suturing and tension‐free closure; (5) postoperative care, including plaque control and slow re-entry of function. In the cited study, postoperative evaluations showed consistent gains at 12 months, and patient comfort was favorable. Success hinges on graft adaptation to the bed, hemostasis, and removal of movement, given that the “dead skin” scaffold must integrate before functional loading.
5. Advantages and Limitations of the use of dead skin in developing new gingiva
Advantages of the use of dead skin in developing new gingiva include: reduced donor‐site morbidity (no palatal graft harvest), predictable gain in keratinized tissue width, and good patient tolerance as shown in clinical trials. The donor tissue (skin allograft) offers a ready‐to‐use, standardized scaffold. On the limitation side: cost and availability of processed skin allograft tissue, potential risks of immunogenic response (though minimal with devitalized tissue), and the need for long-term outcome studies beyond 12 months. Moreover, the approach remains less widespread in periodontal practice compared to autogenous grafts such as connective tissue grafts. Specialists should weigh these factors when selecting the treatment plan.
6. Future Directions: Enhancing the use of dead skin in developing new gingiva
Turning to future directions, the concept of the use of dead skin in developing new gingiva is ripe for refinement. Emerging tissue‐engineering combinations—such as skin allograft scaffolds seeded with autologous gingival stem cells (GMSCs), or augmented with growth‐factor delivery systems—could further enhance outcomes. Additionally, biomaterials research is exploring optimized scaffolds that mimic both skin and gingiva extracellular matrices. For example, studies highlight the basement membrane and dermal‐epidermal junction roles in regeneration. Future clinical trials comparing skin allograft vs autogenous graft vs xenograft will help refine protocols, cost-effectiveness, and long-term stability in soft‐tissue augmentation for implants or periodontal therapy.
Frequently Asked Questions (FAQs)
Q1: Is the use of dead skin in developing new gingiva safe?
A1: Yes — clinical trials (e.g., freeze-dried skin allograft) have demonstrated acceptable safety profiles with no major sequelae at 12 months. As always, donor‐tissue screening, consent, and infection control are paramount.
Q2: How much keratinized gingiva gain can be expected?
A2: One study reported a mean gain of approximately 2.88 mm (±1.57 mm) at 12 months using a skin allograft. Results depend on initial defect size, site preparation, and post-operative compliance.
Q3: Does the use of dead skin in developing new gingiva replace the palatal graft harvest?
A3: It offers a viable alternative when palatal harvest is contraindicated or the patient prefers a less invasive option. However, autogenous grafts remain the gold standard in many cases.
Q4: What is the healing time compared to conventional grafts?
A4: Healing appears similar or slightly faster, given the gingiva’s innate regenerative capacity and favorable integration of the skin scaffold. Studies show host tissue integration at 12 months with minimal complications.
Q5: Are there contraindications for the use of dead skin in developing new gingiva?
A5: Yes. Contraindications include patients with uncontrolled systemic disease, smokers with poor compliance, sites with inadequate vascular bed, and situations where donor tissue allograft is contraindicated or not available.
In conclusion, the use of dead skin in developing new gingiva represents a sophisticated tool in the armamentarium of periodontal and implant‐soft tissue specialists. As research deepens and protocols refine, it stands to offer a less invasive, patient-friendly alternative to conventional grafts—while achieving predictable gains in keratinized attached gingiva, enhancing soft-tissue management around teeth and implants, and supporting long-term periodontal stability.
References
Gingival-derived mesenchymal stem cells: An endless resource for regenerative dentistry. PMC.
(the basement membrane in epidermal polarity, stemness, and regeneration). arXiv.
Freeze-dried skin allografts. A human clinical and histological study. PubMed.
The importance of keratinized gingiva surrounding dental implants. (MedCrave online)
A Cutting-Edge Perspective for Dental Specialists
As periodontal and gingival surgery expands into regenerative frontiers, the concept of incorporating materials traditionally reserved for dermatologic or tissue-engineering purposes is gaining traction. One of these materials is the use of dead skin or skin allograft tissue in the development of new gingiva, specifically in augmenting keratinized attached gingiva. This article is tailored for dental specialists and explores how this innovative approach can bolster periodontal outcomes, the biological rationale, clinical evidence, and practical considerations. Key search phrase: use of dead skin in developing new gingiva.
1. Understanding the Biological Basis for the use of dead skin in developing new gingiva
To grasp why the use of dead skin in developing new gingiva might be feasible, one must review the histology of both skin and gingiva. The keratinized gingiva acts as a robust barrier that sustains mechanical insult, bacterial challenge, and shear forces. Skin tissue, similarly keratinized, offers a structural paradigm that can be adapted. In tissue‐engineering studies, skin allografts (for example, freeze-dried skin) have been successfully used to treat mucogingival problems, showing increases in the width of keratinized tissue. By applying the principle of “dead skin” (i.e., processed skin allograft) as a scaffold or grafting medium, clinicians may harness the structural and biological benefits of the skin dermal/epidermal complex to regenerate gingival tissue.
2. Clinical Evidence Supporting the use of dead skin in developing new gingiva
Dental specialists looking at the use of dead skin in developing new gingiva should evaluate the available clinical data. One landmark study describes the use of freeze-dried skin (FDS) allograft in human patients with mucogingival defects: 14 patients received 24 FDS allografts over 56 tooth sites, showing a mean gain of keratinized gingiva of approximately 2.88 ± 1.57 mm at 12 months. These results support that skin allografts may serve as a stable alternative for gingival augmentation. Additionally, comparative research of skin versus gingival keratinocyte healing shows that gingival keratinocytes exhibit faster re-epithelialization. Such data suggest that while gingiva may innately regenerate well, the introduction of a skin-derived scaffold may enhance or accelerate that process, giving credence to the concept of the use of dead skin in developing new gingiva.
3. Mechanisms of Action: When You Apply the use of dead skin in developing new gingiva
The term “use of dead skin in developing new gingiva” refers to the grafting of devitalized skin tissue (epidermal + dermal layers) onto a prepared gingival bed. The graft acts as a scaffold: the dermal matrix supports vascular ingrowth and fibroblast migration; the epidermal layer may provide a keratinized barrier. Over time, host gingival tissues integrate, remodel, and differentiate, establishing a new zone of keratinized attached gingiva. The superior wound-healing properties of gingiva (versus skin) are well-documented. By placing a “dead skin” graft, you harness both the mechanical structure of skin tissue and the regenerative milieu of the gingiva. Importantly, the scaffold is devitalized (hence “dead”), reducing immune reaction while enabling host cell infiltration and remodeling. The result: a predictable band of keratinized gingiva, improved soft-tissue resilience, and enhanced periodontal stability.
4. Indications and Clinical Protocols for the use of dead skin in developing new gingiva
Dental specialists should consider the use of dead skin in developing new gingiva when facing cases such as inadequate keratinized tissue width, recession with minimal attached gingiva, implant sites lacking soft-tissue bulk, or mucogingival defects resistant to conventional grafting. The clinical protocol may involve: (1) patient assessment and consent regarding allograft use; (2) recipient site preparation (partial‐thickness flap, apical periosteal separation); (3) rehydration and placement of the skin allograft; (4) suturing and tension‐free closure; (5) postoperative care, including plaque control and slow re-entry of function. In the cited study, postoperative evaluations showed consistent gains at 12 months, and patient comfort was favorable. Success hinges on graft adaptation to the bed, hemostasis, and removal of movement, given that the “dead skin” scaffold must integrate before functional loading.
5. Advantages and Limitations of the use of dead skin in developing new gingiva
Advantages of the use of dead skin in developing new gingiva include: reduced donor‐site morbidity (no palatal graft harvest), predictable gain in keratinized tissue width, and good patient tolerance as shown in clinical trials. The donor tissue (skin allograft) offers a ready‐to‐use, standardized scaffold. On the limitation side: cost and availability of processed skin allograft tissue, potential risks of immunogenic response (though minimal with devitalized tissue), and the need for long-term outcome studies beyond 12 months. Moreover, the approach remains less widespread in periodontal practice compared to autogenous grafts such as connective tissue grafts. Specialists should weigh these factors when selecting the treatment plan.
6. Future Directions: Enhancing the use of dead skin in developing new gingiva
Turning to future directions, the concept of the use of dead skin in developing new gingiva is ripe for refinement. Emerging tissue‐engineering combinations—such as skin allograft scaffolds seeded with autologous gingival stem cells (GMSCs), or augmented with growth‐factor delivery systems—could further enhance outcomes. Additionally, biomaterials research is exploring optimized scaffolds that mimic both skin and gingiva extracellular matrices. For example, studies highlight the basement membrane and dermal‐epidermal junction roles in regeneration. Future clinical trials comparing skin allograft vs autogenous graft vs xenograft will help refine protocols, cost-effectiveness, and long-term stability in soft‐tissue augmentation for implants or periodontal therapy.
Frequently Asked Questions (FAQs)
Q1: Is the use of dead skin in developing new gingiva safe?
A1: Yes — clinical trials (e.g., freeze-dried skin allograft) have demonstrated acceptable safety profiles with no major sequelae at 12 months. As always, donor‐tissue screening, consent, and infection control are paramount.
Q2: How much keratinized gingiva gain can be expected?
A2: One study reported a mean gain of approximately 2.88 mm (±1.57 mm) at 12 months using a skin allograft. Results depend on initial defect size, site preparation, and post-operative compliance.
Q3: Does the use of dead skin in developing new gingiva replace the palatal graft harvest?
A3: It offers a viable alternative when palatal harvest is contraindicated or the patient prefers a less invasive option. However, autogenous grafts remain the gold standard in many cases.
Q4: What is the healing time compared to conventional grafts?
A4: Healing appears similar or slightly faster, given the gingiva’s innate regenerative capacity and favorable integration of the skin scaffold. Studies show host tissue integration at 12 months with minimal complications.
Q5: Are there contraindications for the use of dead skin in developing new gingiva?
A5: Yes. Contraindications include patients with uncontrolled systemic disease, smokers with poor compliance, sites with inadequate vascular bed, and situations where donor tissue allograft is contraindicated or not available.
In conclusion, the use of dead skin in developing new gingiva represents a sophisticated tool in the armamentarium of periodontal and implant‐soft tissue specialists. As research deepens and protocols refine, it stands to offer a less invasive, patient-friendly alternative to conventional grafts—while achieving predictable gains in keratinized attached gingiva, enhancing soft-tissue management around teeth and implants, and supporting long-term periodontal stability.
References
Gingival-derived mesenchymal stem cells: An endless resource for regenerative dentistry. PMC.
(the basement membrane in epidermal polarity, stemness, and regeneration). arXiv.
Freeze-dried skin allografts. A human clinical and histological study. PubMed.
The importance of keratinized gingiva surrounding dental implants. (MedCrave online)
Magnetically Guided Dental Nanobots (CalBots) Are Real
Magnetically Guided Dental Nanobots (CalBots) Are Real The concept of nanobots in dentistry has long been associated with futuristic speculation. However, recent peer-reviewed research [...]
Peri-Implantitis Treatment & Prevention Methods
Peri-Implantitis Treatment & Prevention Methods Peri-implantitis remains one of the most significant biological complications affecting dental implants, posing a serious threat to long-term implant [...]
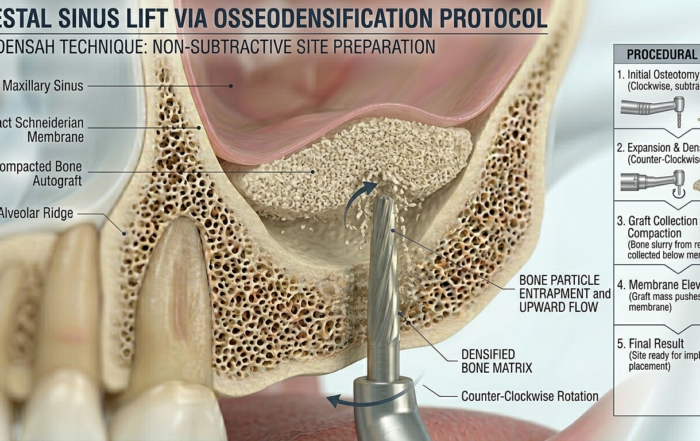
Latest Sinus Lifting Techniques in Modern Implant Dentistry
Latest Sinus Lifting Techniques in Modern Implant Dentistry A Comprehensive Clinical Review Introduction to Sinus Lifting in Implantology Sinus lifting, or maxillary sinus floor [...]
Biomechanics of the Triple Abutment & BOPiT Concept
Dental Biomechanics · Implant Science · Clinical Evidence Biomechanics of the Triple Abutment & BOPiT Concept How a saddle-shaped mathematical surface is rewriting the rules of load distribution [...]
The Woman Who Proved One Implant Could Hold Three Crowns
The Woman Who Proved One Implant Could Hold Three Crowns While the dental establishment looked away, Dr. Luciana Colepícolo spent 12 years building the [...]
ENPP1, The Molecular Brake That Limits Tissue Healing
ENPP1, The Molecular Brake That Limits Tissue Healing Introduction: A New Biological Barrier to Dental Regeneration Meet ENPP1—a protein most dentists have never heard [...]