Supramolecular Plastics That Disappear in Seawater: A Breakthrough for a Cleaner Planet
In a remarkable scientific breakthrough, researchers have created a new kind of supramolecular plastics that can completely dissolve in seawater within hours. Developed by a team from Japan’s RIKEN Center for Emergent Matter Science and the University of Tokyo, led by Professor Takuzo Aida, this material marks a revolutionary step in combating the global plastic pollution crisis. With the oceans increasingly burdened by millions of tons of non-degradable waste, this discovery offers a practical, scalable, and sustainable solution to restore marine health and protect biodiversity.
Who Created the Supramolecular Plastic?
The innovation stems from a collaboration between RIKEN and the University of Tokyo, spearheaded by Professor Takuzo Aida — a world leader in supramolecular chemistry and polymer science. The team’s goal was to design a new class of biodegradable plastics that match the mechanical strength of conventional materials while being environmentally safe. By leveraging reversible molecular interactions rather than permanent covalent bonds, they succeeded in developing a plastic that’s both durable during use and capable of vanishing harmlessly in seawater. This marks a transformative step in the field of eco-friendly polymer materials.
What Makes Supramolecular Plastics Unique?
Unlike petroleum-based plastics made from hydrocarbons, this new material is composed of supramolecular polymers held together by reversible salt bridges. These ionic interactions give the material exceptional strength under dry or freshwater conditions but allow it to disintegrate when exposed to the salt ions in seawater. The design takes cues from nature’s dynamic molecular systems, where weak interactions can build strong yet adaptable structures. The result is a biodegradable plastic that maintains stability during use, yet fully dissolves in marine environments — leaving no microplastics or chemical residues behind.
How Do Supramolecular Plastics Dissolve?
The mechanism behind this innovation lies in the material’s molecular architecture. The plastic is formed by mixing two ionic monomers — sodium hexametaphosphate and guanidinium-based compounds. In regular conditions, these monomers are bound by strong electrostatic forces that create a flexible yet stable plastic. However, when the material comes into contact with seawater, the abundant salt ions disrupt these electrostatic interactions. The polymer structure disassembles naturally, allowing the entire plastic to dissolve completely within hours. No microplastic fragments or toxic byproducts are produced — a crucial difference from conventional “biodegradable” plastics that still pollute marine ecosystems.
Environmental and Health Benefits of Supramolecular Plastics
This new class of supramolecular plastics offers far-reaching environmental and health benefits. First and foremost, it is non-toxic, carbon-neutral, and flame-retardant. It does not emit carbon dioxide during decomposition, minimizing its carbon footprint. Once dissolved, its components can be safely digested by marine bacteria, effectively closing the ecological loop. Because it doesn’t require industrial composting or specialized recycling facilities, this material could revolutionize global waste management — particularly in regions lacking advanced infrastructure. From reducing ocean pollution to safeguarding marine biodiversity, supramolecular plastics represent a truly sustainable approach to material science.
Future Potential and Urgency
Global plastic pollution is projected to triple by 2040 unless urgent action is taken. This alarming trend underscores the necessity for groundbreaking innovations like seawater-dissolving supramolecular plastics. The RIKEN team is already developing functional coatings, films, and packaging applications using this technology. Imagine fishing nets, food packaging, or shipping materials that retain strength during use but vanish once discarded into the ocean. Such innovations could drastically cut the amount of plastic waste accumulating in our seas, offering a realistic and scalable pathway toward a circular and sustainable materials economy.
Integrating Biomimetic and Supramolecular Design
The foundation of this breakthrough lies in the integration of biomimetic and supramolecular design principles. Nature’s systems — from proteins to cell membranes — rely on reversible molecular interactions to maintain balance between stability and adaptability. By applying similar principles, the researchers designed a plastic that behaves like biological material: strong under normal conditions, yet capable of disassembly when environmental conditions change. This approach has opened a new frontier in materials engineering, demonstrating how molecular-level control can lead to functional, sustainable, and recyclable materials that align with the planet’s ecological needs.
Advantages Over Conventional Plastics
Compared to traditional petroleum-derived polymers, supramolecular plastics offer a host of advantages. They can be manufactured under mild conditions, reducing energy consumption and eliminating the dependence on fossil fuels. Their reversible bonding nature enables recycling without structural degradation, ensuring a longer and more efficient lifecycle. Furthermore, their flame-retardant and thermally stable properties make them suitable replacements for single-use plastics in multiple industries, including packaging, consumer goods, and textiles. In short, supramolecular plastics bridge the gap between performance and sustainability — something conventional plastics have never achieved.
Challenges and Next Steps
While promising, the road to commercialization is still in progress. Researchers are now optimizing large-scale production methods to ensure consistent performance and affordability. Balancing the plastic’s stability during use with its rapid dissolvability in seawater remains a key focus. Further research is also exploring medical and industrial adaptations, where controlled degradability could be an asset. With continued support from global environmental policies and industry collaboration, supramolecular plastics could soon move from the laboratory to real-world applications — reshaping how we manufacture, use, and dispose of materials.
Real-World Impact and Global Collaboration
The creation of seawater-dissolving supramolecular plastics highlights the importance of international cooperation in solving environmental crises. Japan’s leadership through institutions like RIKEN and the University of Tokyo showcases how cutting-edge science can produce tangible sustainability solutions. As governments, researchers, and industries align on the urgent need for cleaner materials, this discovery could catalyze a global transition toward biodegradable, recyclable, and ocean-safe polymers. It’s not merely a scientific success — it’s a roadmap for a greener, circular economy.
Is There a Place for This Invention in Dental Practice?
Although this innovation was primarily developed for environmental applications, its potential extends into healthcare and dental materials. Dentistry contributes significantly to global plastic waste through disposable items such as impression trays, packaging, and barrier films. If adapted for medical-grade use, biodegradable supramolecular plastics could offer eco-friendly alternatives that dissolve harmlessly after disposal. Temporary dental devices, sterile films, or protective packaging could all benefit from this self-degrading chemistry — dramatically reducing the environmental footprint of modern dental practice. As safety and sterilization standards evolve, the dental industry may well adopt these materials as part of its shift toward sustainable healthcare solutions.
Broader Implications for Sustainability
The emergence of supramolecular plastics represents a major leap in sustainable material design. By creating polymers that “know when to disappear,” scientists are redefining the role of plastics in society. These materials transform plastic from a pollutant into a temporary, responsible tool — strong and useful during its life, yet harmless after its purpose is served. This innovation, rooted in biomimetic chemistry and molecular engineering, embodies the future of eco-conscious technology. With continued research and collaboration, supramolecular plastics could usher in a new era of environmental responsibility — where science and sustainability go hand in hand.
FAQs About Supramolecular Plastics That Dissolve in Seawater
1. How long does it take for supramolecular plastic to dissolve in seawater?
Under normal marine conditions, the plastic dissolves completely within a few hours, leaving no microplastics or toxic residues behind.
2. Is the plastic safe for marine life?
Yes. Once dissolved, the molecular components are biodegradable and non-toxic, allowing natural marine bacteria to metabolize them safely.
3. Can this plastic be used for everyday packaging?
The research team is developing coatings and films suitable for packaging, particularly for items prone to ocean exposure, such as food wrappers and fishing materials.
4. How do supramolecular plastics compare to regular biodegradable plastics?
Traditional biodegradable plastics often need industrial composting and can still release microplastics. Supramolecular plastics dissolve naturally and completely in seawater.
5. When will this material be commercially available?
Commercial applications are under development, with scalable production expected within the next decade as manufacturing technologies advance.
Reference
Aida, T. et al. (2024). *“A Supramolecular Plastic That Dissolves in Seawater within Hours.”* Nature Communications, RIKEN Center for Emergent Matter Science and University of Tokyo.
Reference
Mechanically strong yet metabolizable supramolecular plastics by desalting upon phase separation
Yiren Cheng, Eiji Hirano, Hao Wang, Motonobu Kuwayama, E. W. Meijer, Hubiao Huang, and Takuzo Aida
Recent Articles
In a remarkable scientific breakthrough, researchers have created a new kind of supramolecular plastics that can completely dissolve in seawater within hours. Developed by a team from Japan’s RIKEN Center for Emergent Matter Science and the University of Tokyo, led by Professor Takuzo Aida, this material marks a revolutionary step in combating the global plastic pollution crisis. With the oceans increasingly burdened by millions of tons of non-degradable waste, this discovery offers a practical, scalable, and sustainable solution to restore marine health and protect biodiversity.
Who Created the Supramolecular Plastic?
The innovation stems from a collaboration between RIKEN and the University of Tokyo, spearheaded by Professor Takuzo Aida — a world leader in supramolecular chemistry and polymer science. The team’s goal was to design a new class of biodegradable plastics that match the mechanical strength of conventional materials while being environmentally safe. By leveraging reversible molecular interactions rather than permanent covalent bonds, they succeeded in developing a plastic that’s both durable during use and capable of vanishing harmlessly in seawater. This marks a transformative step in the field of eco-friendly polymer materials.
What Makes Supramolecular Plastics Unique?
Unlike petroleum-based plastics made from hydrocarbons, this new material is composed of supramolecular polymers held together by reversible salt bridges. These ionic interactions give the material exceptional strength under dry or freshwater conditions but allow it to disintegrate when exposed to the salt ions in seawater. The design takes cues from nature’s dynamic molecular systems, where weak interactions can build strong yet adaptable structures. The result is a biodegradable plastic that maintains stability during use, yet fully dissolves in marine environments — leaving no microplastics or chemical residues behind.
How Do Supramolecular Plastics Dissolve?
The mechanism behind this innovation lies in the material’s molecular architecture. The plastic is formed by mixing two ionic monomers — sodium hexametaphosphate and guanidinium-based compounds. In regular conditions, these monomers are bound by strong electrostatic forces that create a flexible yet stable plastic. However, when the material comes into contact with seawater, the abundant salt ions disrupt these electrostatic interactions. The polymer structure disassembles naturally, allowing the entire plastic to dissolve completely within hours. No microplastic fragments or toxic byproducts are produced — a crucial difference from conventional “biodegradable” plastics that still pollute marine ecosystems.
Environmental and Health Benefits of Supramolecular Plastics
This new class of supramolecular plastics offers far-reaching environmental and health benefits. First and foremost, it is non-toxic, carbon-neutral, and flame-retardant. It does not emit carbon dioxide during decomposition, minimizing its carbon footprint. Once dissolved, its components can be safely digested by marine bacteria, effectively closing the ecological loop. Because it doesn’t require industrial composting or specialized recycling facilities, this material could revolutionize global waste management — particularly in regions lacking advanced infrastructure. From reducing ocean pollution to safeguarding marine biodiversity, supramolecular plastics represent a truly sustainable approach to material science.
Future Potential and Urgency
Global plastic pollution is projected to triple by 2040 unless urgent action is taken. This alarming trend underscores the necessity for groundbreaking innovations like seawater-dissolving supramolecular plastics. The RIKEN team is already developing functional coatings, films, and packaging applications using this technology. Imagine fishing nets, food packaging, or shipping materials that retain strength during use but vanish once discarded into the ocean. Such innovations could drastically cut the amount of plastic waste accumulating in our seas, offering a realistic and scalable pathway toward a circular and sustainable materials economy.
Integrating Biomimetic and Supramolecular Design
The foundation of this breakthrough lies in the integration of biomimetic and supramolecular design principles. Nature’s systems — from proteins to cell membranes — rely on reversible molecular interactions to maintain balance between stability and adaptability. By applying similar principles, the researchers designed a plastic that behaves like biological material: strong under normal conditions, yet capable of disassembly when environmental conditions change. This approach has opened a new frontier in materials engineering, demonstrating how molecular-level control can lead to functional, sustainable, and recyclable materials that align with the planet’s ecological needs.
Advantages Over Conventional Plastics
Compared to traditional petroleum-derived polymers, supramolecular plastics offer a host of advantages. They can be manufactured under mild conditions, reducing energy consumption and eliminating the dependence on fossil fuels. Their reversible bonding nature enables recycling without structural degradation, ensuring a longer and more efficient lifecycle. Furthermore, their flame-retardant and thermally stable properties make them suitable replacements for single-use plastics in multiple industries, including packaging, consumer goods, and textiles. In short, supramolecular plastics bridge the gap between performance and sustainability — something conventional plastics have never achieved.
Challenges and Next Steps
While promising, the road to commercialization is still in progress. Researchers are now optimizing large-scale production methods to ensure consistent performance and affordability. Balancing the plastic’s stability during use with its rapid dissolvability in seawater remains a key focus. Further research is also exploring medical and industrial adaptations, where controlled degradability could be an asset. With continued support from global environmental policies and industry collaboration, supramolecular plastics could soon move from the laboratory to real-world applications — reshaping how we manufacture, use, and dispose of materials.
Real-World Impact and Global Collaboration
The creation of seawater-dissolving supramolecular plastics highlights the importance of international cooperation in solving environmental crises. Japan’s leadership through institutions like RIKEN and the University of Tokyo showcases how cutting-edge science can produce tangible sustainability solutions. As governments, researchers, and industries align on the urgent need for cleaner materials, this discovery could catalyze a global transition toward biodegradable, recyclable, and ocean-safe polymers. It’s not merely a scientific success — it’s a roadmap for a greener, circular economy.
Is There a Place for This Invention in Dental Practice?
Although this innovation was primarily developed for environmental applications, its potential extends into healthcare and dental materials. Dentistry contributes significantly to global plastic waste through disposable items such as impression trays, packaging, and barrier films. If adapted for medical-grade use, biodegradable supramolecular plastics could offer eco-friendly alternatives that dissolve harmlessly after disposal. Temporary dental devices, sterile films, or protective packaging could all benefit from this self-degrading chemistry — dramatically reducing the environmental footprint of modern dental practice. As safety and sterilization standards evolve, the dental industry may well adopt these materials as part of its shift toward sustainable healthcare solutions.
Broader Implications for Sustainability
The emergence of supramolecular plastics represents a major leap in sustainable material design. By creating polymers that “know when to disappear,” scientists are redefining the role of plastics in society. These materials transform plastic from a pollutant into a temporary, responsible tool — strong and useful during its life, yet harmless after its purpose is served. This innovation, rooted in biomimetic chemistry and molecular engineering, embodies the future of eco-conscious technology. With continued research and collaboration, supramolecular plastics could usher in a new era of environmental responsibility — where science and sustainability go hand in hand.
FAQs About Supramolecular Plastics That Dissolve in Seawater
1. How long does it take for supramolecular plastic to dissolve in seawater?
Under normal marine conditions, the plastic dissolves completely within a few hours, leaving no microplastics or toxic residues behind.
2. Is the plastic safe for marine life?
Yes. Once dissolved, the molecular components are biodegradable and non-toxic, allowing natural marine bacteria to metabolize them safely.
3. Can this plastic be used for everyday packaging?
The research team is developing coatings and films suitable for packaging, particularly for items prone to ocean exposure, such as food wrappers and fishing materials.
4. How do supramolecular plastics compare to regular biodegradable plastics?
Traditional biodegradable plastics often need industrial composting and can still release microplastics. Supramolecular plastics dissolve naturally and completely in seawater.
5. When will this material be commercially available?
Commercial applications are under development, with scalable production expected within the next decade as manufacturing technologies advance.
Reference
Aida, T. et al. (2024). *“A Supramolecular Plastic That Dissolves in Seawater within Hours.”* Nature Communications, RIKEN Center for Emergent Matter Science and University of Tokyo.
Reference
Mechanically strong yet metabolizable supramolecular plastics by desalting upon phase separation
Yiren Cheng, Eiji Hirano, Hao Wang, Motonobu Kuwayama, E. W. Meijer, Hubiao Huang, and Takuzo Aida
Recent Articles
Peri-Implantitis Treatment & Prevention Methods
Peri-Implantitis Treatment & Prevention Methods Peri-implantitis remains one of the most significant biological complications affecting dental implants, posing a serious threat to long-term implant [...]
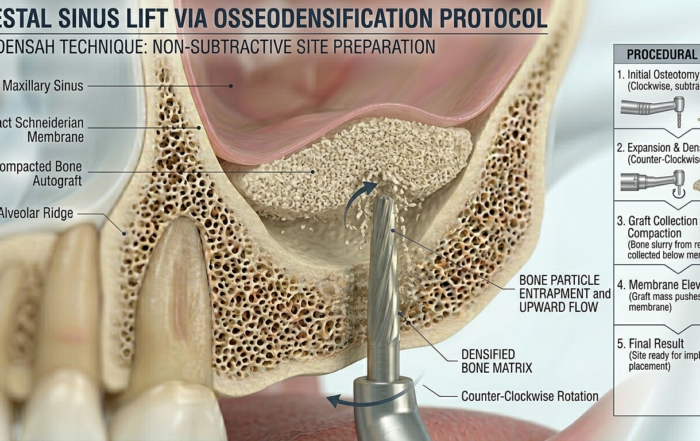
Latest Sinus Lifting Techniques in Modern Implant Dentistry
Latest Sinus Lifting Techniques in Modern Implant Dentistry A Comprehensive Clinical Review Introduction to Sinus Lifting in Implantology Sinus lifting, or maxillary sinus floor [...]
Biomechanics of the Triple Abutment & BOPiT Concept
Dental Biomechanics · Implant Science · Clinical Evidence Biomechanics of the Triple Abutment & BOPiT Concept How a saddle-shaped mathematical surface is rewriting the rules of load distribution [...]
The Woman Who Proved One Implant Could Hold Three Crowns
The Woman Who Proved One Implant Could Hold Three Crowns While the dental establishment looked away, Dr. Luciana Colepícolo spent 12 years building the [...]
ENPP1, The Molecular Brake That Limits Tissue Healing
ENPP1, The Molecular Brake That Limits Tissue Healing Introduction: A New Biological Barrier to Dental Regeneration Meet ENPP1—a protein most dentists have never heard [...]
The Rise of Self‑Healing Dental Biomaterials
The Rise of Self‑Healing Dental Biomaterials Introduction In modern restorative dentistry, durability and longevity of materials remain among the greatest clinical challenges. Traditional dental [...]