The Future of Smiles: Breakthroughs in Enamel Regeneration You Need to Know About
Enamel Regeneration: Why Can’t Our Teeth Heal Themselves?
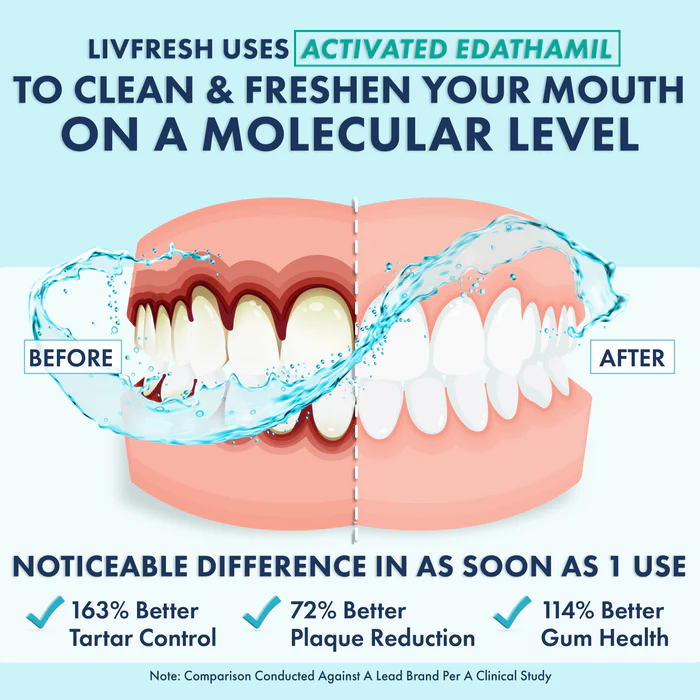
Imagine if your teeth could fix themselves. Like a lizard regrowing its tail or your skin healing from a scrape, enamel regeneration sounds like science fiction. But here’s the reality: over 90% of adults experience enamel loss, and the human body simply can’t regenerate it. Why? Because the specialized enamel-producing cells, ” ameloblasts”, disappear once your teeth erupt.
Enter a new era of regenerative dentistry, where scientists are harnessing stem cells, bioengineering, and computer-designed proteins to regrow dental enamel.
The Quest for Enamel Regeneration Starts With Stem Cells
Regenerating enamel starts by recreating ameloblasts, the powerhouse cells responsible for building enamel. Scientists at the University of Washington and Harvard have made big strides by using human induced pluripotent stem cells (hiPSCs) to create induced ameloblasts (iAMs).
But there was a catch: these iAMs wouldn’t mature into their enamel-spitting final form without a little help, usually from their neighboring cells, odontoblasts, the cells responsible for forming dentin. The mystery was why this cellular teamwork was necessary.
Cracking the Code: Notch Signaling and Tooth Cell Chatter
In a plot twist worthy of a dental drama, the researchers uncovered the secret handshake between these tooth cells: a communication system called the Notch pathway.
Normally, this pathway needs physical contact between cells. Odontoblasts use proteins like Delta-like ligands to activate Notch receptors on ameloblasts, nudging them into maturity. This discovery was a big deal—it revealed how enamel-building instructions are passed along at the cellular level.
Say Hello to C3-DLL4: The AI-Designed Protein Rewriting Dental History
Here’s where the science gets seriously futuristic.
The research team designed a soluble Notch activator called C3-DLL4 , a computer-crafted protein that mimics the natural Delta ligand but doesn’t require physical contact between cells. When added to iAM organoids (little lab-grown tooth cell balls), C3-DLL4 successfully matured them into enamel-secreting ameloblasts, even without odontoblasts around.
Yes, you read that right—they got enamel-producing cells to develop in isolation, just by activating the right signals with a lab-made molecule. It’s like giving a plant the exact sunlight and water recipe it needs to bloom—no soil needed.
DLX3 : The Gene That Holds the Keys to Enamel Formation
The team also explored the role of a gene called DLX3, known to cause Amelogenesis Imperfecta when mutated. Using CRISPR (yes, the gene-editing tech that’s changing medicine), they knocked out DLX3 in stem cells and watched what happened.
Turns out, DLX3 isn’t needed for starting ameloblast formation—but it is critical for their final maturation and the production of enamel proteins like Enamelin and MMP20.
This means DLX3 acts like the “finishing school” teacher for ameloblasts—guiding them through the final stage of their transformation into enamel-secreting machines.
Why This Enamel Regeneration Research Matters to You
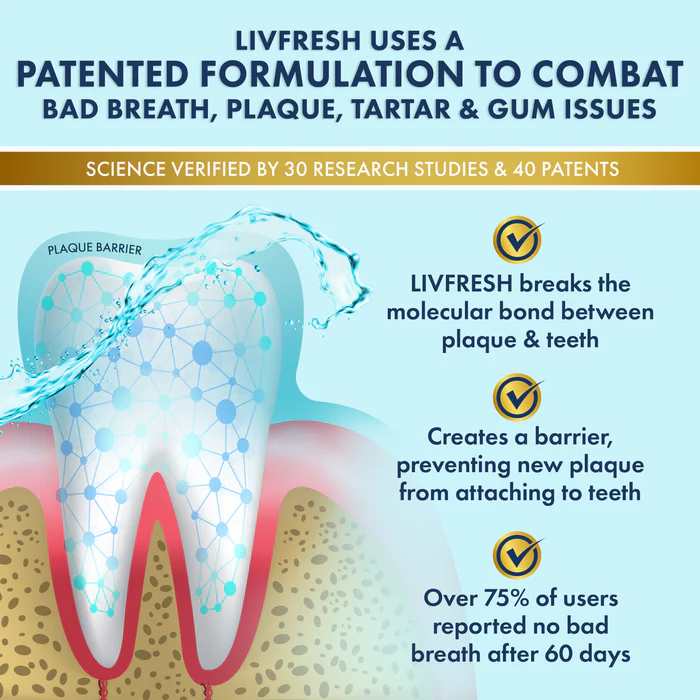
If you’re a dentist, this research is pure gold. It paves the way for cell-based therapies to repair or regenerate damaged enamel, a game-changer for patients suffering from erosion, cavities, or genetic enamel disorders.
If you’re a patient dreaming of a cavity-proof future, this means you might one day regrow your enamel naturally—no drilling required.
What’s Next for Enamel Regeneration?
While we’re not yet ready to toss out fillings and crowns, this research brings us closer to clinical applications in enamel repair. The next steps? Translating this lab success into real-world therapies, ensuring safety, and figuring out how to deliver these supercharged stem cells or proteins directly to your teeth.
Enamel Regeneration Is No Longer Just a Dream
From the discovery of Notch’s pivotal role to the invention of a smart, soluble activator that turns stem cells into enamel architects, the future of dental care is glowing with potential. Enamel regeneration is on the horizon, and your smile might just be the next to benefit.
Meet Dr. Hannele Ruohola-Baker
Dr. Hannele Ruohola-Baker is a Finnish-American biochemist and a leading figure in stem cell research. She serves as a professor of biochemistry and the associate director of the Institute for Stem Cell and Regenerative Medicine at the University of Washington. Her research focuses on the molecular biology of stem cells and the use of model organisms like Drosophila to study human diseases.

Enamel Regeneration – Dr Hannele Ruohola-Baker
In recent advancements, Dr. Ruohola-Baker’s laboratory has made significant strides in dental regenerative medicine. Her team successfully developed tooth organoids from human stem cells that secrete key enamel proteins, marking a pivotal step toward bioengineered solutions for tooth repair and regeneration. This research aims to pave the way for “living fillings” that could naturally restore tooth enamel, potentially transforming dental care practices.
Dr. Ruohola-Baker envisions a future where regenerative dentistry becomes a reality, offering innovative treatments for oral health challenges.
Reference:
Recent Articles
Enamel Regeneration: Why Can’t Our Teeth Heal Themselves?
Imagine if your teeth could fix themselves. Like a lizard regrowing its tail or your skin healing from a scrape, enamel regeneration sounds like science fiction. But here’s the reality: over 90% of adults experience enamel loss, and the human body simply can’t regenerate it. Why? Because the specialized enamel-producing cells, ” ameloblasts”, disappear once your teeth erupt.
Enter a new era of regenerative dentistry, where scientists are harnessing stem cells, bioengineering, and computer-designed proteins to regrow dental enamel.
The Quest for Enamel Regeneration Starts With Stem Cells
Regenerating enamel starts by recreating ameloblasts, the powerhouse cells responsible for building enamel. Scientists at the University of Washington and Harvard have made big strides by using human induced pluripotent stem cells (hiPSCs) to create induced ameloblasts (iAMs).
But there was a catch: these iAMs wouldn’t mature into their enamel-spitting final form without a little help, usually from their neighboring cells, odontoblasts, the cells responsible for forming dentin. The mystery was why this cellular teamwork was necessary.
Cracking the Code: Notch Signaling and Tooth Cell Chatter
In a plot twist worthy of a dental drama, the researchers uncovered the secret handshake between these tooth cells: a communication system called the Notch pathway.
Normally, this pathway needs physical contact between cells. Odontoblasts use proteins like Delta-like ligands to activate Notch receptors on ameloblasts, nudging them into maturity. This discovery was a big deal—it revealed how enamel-building instructions are passed along at the cellular level.
Say Hello to C3-DLL4: The AI-Designed Protein Rewriting Dental History
Here’s where the science gets seriously futuristic.
The research team designed a soluble Notch activator called C3-DLL4 , a computer-crafted protein that mimics the natural Delta ligand but doesn’t require physical contact between cells. When added to iAM organoids (little lab-grown tooth cell balls), C3-DLL4 successfully matured them into enamel-secreting ameloblasts, even without odontoblasts around.
Yes, you read that right—they got enamel-producing cells to develop in isolation, just by activating the right signals with a lab-made molecule. It’s like giving a plant the exact sunlight and water recipe it needs to bloom—no soil needed.
DLX3 : The Gene That Holds the Keys to Enamel Formation
The team also explored the role of a gene called DLX3, known to cause Amelogenesis Imperfecta when mutated. Using CRISPR (yes, the gene-editing tech that’s changing medicine), they knocked out DLX3 in stem cells and watched what happened.
Turns out, DLX3 isn’t needed for starting ameloblast formation—but it is critical for their final maturation and the production of enamel proteins like Enamelin and MMP20.
This means DLX3 acts like the “finishing school” teacher for ameloblasts—guiding them through the final stage of their transformation into enamel-secreting machines.
Why This Enamel Regeneration Research Matters to You
If you’re a dentist, this research is pure gold. It paves the way for cell-based therapies to repair or regenerate damaged enamel, a game-changer for patients suffering from erosion, cavities, or genetic enamel disorders.
If you’re a patient dreaming of a cavity-proof future, this means you might one day regrow your enamel naturally—no drilling required.
What’s Next for Enamel Regeneration?
While we’re not yet ready to toss out fillings and crowns, this research brings us closer to clinical applications in enamel repair. The next steps? Translating this lab success into real-world therapies, ensuring safety, and figuring out how to deliver these supercharged stem cells or proteins directly to your teeth.
Enamel Regeneration Is No Longer Just a Dream
From the discovery of Notch’s pivotal role to the invention of a smart, soluble activator that turns stem cells into enamel architects, the future of dental care is glowing with potential. Enamel regeneration is on the horizon, and your smile might just be the next to benefit.
Meet Dr. Hannele Ruohola-Baker
Dr. Hannele Ruohola-Baker is a Finnish-American biochemist and a leading figure in stem cell research. She serves as a professor of biochemistry and the associate director of the Institute for Stem Cell and Regenerative Medicine at the University of Washington. Her research focuses on the molecular biology of stem cells and the use of model organisms like Drosophila to study human diseases.

Enamel Regeneration – Dr Hannele Ruohola-Baker
In recent advancements, Dr. Ruohola-Baker’s laboratory has made significant strides in dental regenerative medicine. Her team successfully developed tooth organoids from human stem cells that secrete key enamel proteins, marking a pivotal step toward bioengineered solutions for tooth repair and regeneration. This research aims to pave the way for “living fillings” that could naturally restore tooth enamel, potentially transforming dental care practices.
Dr. Ruohola-Baker envisions a future where regenerative dentistry becomes a reality, offering innovative treatments for oral health challenges.
Reference:
Recent Articles
Magnetically Guided Dental Nanobots (CalBots) Are Real
Magnetically Guided Dental Nanobots (CalBots) Are Real The concept of nanobots in dentistry has long been associated with futuristic speculation. However, recent peer-reviewed research [...]
Peri-Implantitis Treatment & Prevention Methods
Peri-Implantitis Treatment & Prevention Methods Peri-implantitis remains one of the most significant biological complications affecting dental implants, posing a serious threat to long-term implant [...]
Latest Sinus Lifting Techniques in Modern Implant Dentistry
Latest Sinus Lifting Techniques in Modern Implant Dentistry A Comprehensive Clinical Review Introduction to Sinus Lifting in Implantology Sinus lifting, or maxillary sinus floor [...]
Biomechanics of the Triple Abutment & BOPiT Concept
Dental Biomechanics · Implant Science · Clinical Evidence Biomechanics of the Triple Abutment & BOPiT Concept How a saddle-shaped mathematical surface is rewriting the rules of load distribution [...]
The Woman Who Proved One Implant Could Hold Three Crowns
The Woman Who Proved One Implant Could Hold Three Crowns While the dental establishment looked away, Dr. Luciana Colepícolo spent 12 years building the [...]
ENPP1, The Molecular Brake That Limits Tissue Healing
ENPP1, The Molecular Brake That Limits Tissue Healing Introduction: A New Biological Barrier to Dental Regeneration Meet ENPP1—a protein most dentists have never heard [...]