Meet SinusFuse: The World’s First Sinus Lifting Implant
Dental implantology has entered a new era with the introduction of SinusFuse, a groundbreaking innovation recognized as the world’s first true sinus implant. Designed to address one of the most complex challenges in implant dentistry—insufficient bone height in the posterior maxilla—SinusFuse represents a major technological leap, combining advanced biomechanics, minimally invasive principles, and patented engineering into a single implant system.
Developed under the OsseoFuse Dental Implant System, SinusFuse is not merely an incremental improvement. It is a paradigm shift that redefines how clinicians approach sinus elevation, bone management, and implant placement in compromised maxillary cases.
The Clinical Challenge of Maxillary Sinus Implants
Posterior maxillary implant placement is often complicated by pneumatization of the maxillary sinus and reduced residual bone height following tooth loss. Traditionally, these cases require additional surgical interventions such as lateral window sinus lifts or crestal sinus elevation with grafting materials. While effective, these procedures increase treatment time, cost, patient morbidity, and clinical risk.
SinusFuse was engineered specifically to overcome these limitations by integrating sinus elevation and implant placement into a single, controlled, minimally invasive procedure.
What Is SinusFuse?
SinusFuse is a patented sinus implant within the OsseoFuse Dental Implant System. It is manufactured from high-grade biocompatible titanium and titanium alloys, ensuring excellent osseointegration, mechanical strength, and long-term stability.
Unlike conventional implants, SinusFuse is designed to interact dynamically with the sinus floor. During placement, it performs multiple critical functions simultaneously:
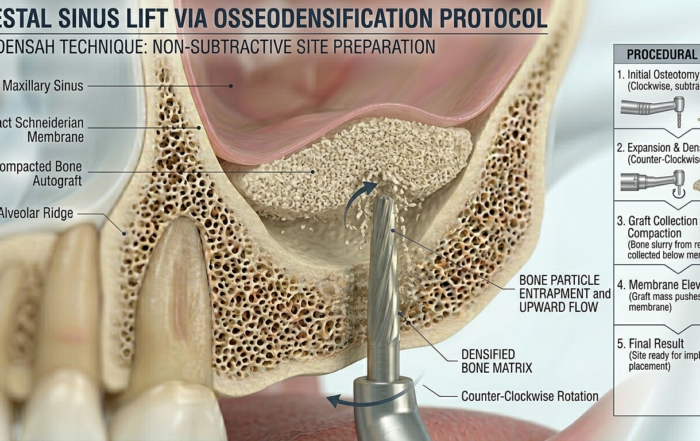
Controlled up-fracture of the sinus floor
Harvesting and transporting autogenous bone
Gentle elevation of the Schneiderian membrane
Immediate implant placement in a single surgical sequence
This integrated functionality eliminates the need for separate sinus lift instruments or grafting procedures in many clinical scenarios.
OsseoFuse Dental Implant System Overview
The OsseoFuse system includes HexaPlus™, ConicalPlus™, and Sinus™ implants, along with compatible abutments, overdenture bars, and restorative components. The system is indicated for use in both the maxilla and mandible, supporting:
Single-tooth restorations
Multiple-unit prostheses
Cement-retained and screw-retained restorations
Overdentures
Fixed bridgework (terminal or intermediate abutments)
The system supports one-stage and two-stage surgical protocols and is intended primarily for delayed loading, allowing predictable osseointegration in a wide range of bone qualities.
Indications for SinusFuse and OsseoFuse Implants
SinusFuse and other OsseoFuse implants are indicated for patients who are partially or fully edentulous, provided adequate bone conditions exist. Minimum bone requirements include:
At least 1 mm of circumferential bone
At least 2 mm of apical bone
They are suitable for cases where traditional implant placement would otherwise require sinus augmentation procedures.
Surgical Planning and Directions for Use
Successful outcomes with SinusFuse rely heavily on proper diagnosis, planning, and surgical execution. Before implant placement, clinicians must evaluate:
Vertical dimension and restorative space
Distance between the alveolar crest and opposing dentition
Abutment selection and final prosthetic design
Importantly, the final prosthesis should be planned before implant placement to ensure restorative-driven implant positioning.
During surgery, continuous sterile irrigation is mandatory to prevent thermal trauma and protect surrounding tissues. Excessive drilling pressure must be avoided, and only sharp, high-quality instruments should be used to preserve bone vitality and enhance osseointegration.
Contraindications and Patient Selection
SinusFuse implants should not be placed in patients with:
Severely insufficient bone volume or density
Poor oral hygiene
Heavy tobacco use
Uncontrolled systemic conditions such as diabetes or blood disorders
Inadequate patient selection or compromised bone conditions significantly increase the risk of implant failure or lack of osseointegration.
Warnings and Precautions
Dental implant surgery is a highly specialized clinical procedure. Proper training in implantology is strongly recommended before using the OsseoFuse system. Incorrect technique may lead to implant instability, excessive bone loss, or restoration failure.
Additional precautions include:
Avoiding electrosurgical devices or lasers near metallic implants
Monitoring for signs of implant mobility, infection, or bone loss
Immediate intervention or removal if implant failure is suspected
Preventing aspiration or swallowing of small components
Avoiding small-diameter implants with angled abutments in posterior regions
The OsseoFuse system has not been evaluated for MRI safety, and compatibility in MR environments has not been established.
Sterility, Cleaning, and Instrument Handling
OsseoFuse implant fixtures, mounts, and cover screws are supplied sterile via gamma irradiation and are ready for use. Abutments, accessories, and surgical instruments are provided non-sterile and must be sterilized before use.
Recommended cleaning protocol includes:
Ultrasonic cleaning for 10 minutes
Manual removal of debris with a soft-bristle brush
Thorough rinsing, including an alcohol rinse to prevent corrosion
Complete air drying
Steam sterilization using validated cycles
These sterilization parameters are validated to a Sterility Assurance Level (SAL) of 10⁻⁶ in accordance with FDA-recognized ISO standards.
Healing, Restoration, and Occlusal Considerations
Healing time varies based on bone quality, tissue response, and surgical assessment. During this period, excessive functional loading must be avoided. Proper occlusal design is essential to prevent overload, which can compromise osseointegration and long-term implant success.
Potential Adverse Events
As with any implant system, potential complications may include:
Failure or loss of osseointegration
Bone dehiscence requiring grafting
Sinus or anatomical structure perforation
Infection, abscess, or inflammation
Persistent pain or altered sensation
Excessive bone loss
Implant fracture
Early diagnosis and intervention are critical to managing these risks effectively.

The Innovator Behind SinusFuse: Dr. C. Leon Chen
Meet the Innovator Behind SinusFuse: Dr. C. Leon Chen
The vision behind SinusFuse belongs to Dr. C. Leon Chen, DMD, MS, DBA, a globally respected authority in implant dentistry and minimally invasive techniques. A graduate of Harvard University School of Dental Medicine and Northwestern University’s Department of Periodontics, Dr. Chen has dedicated over three decades to advancing implant science.
He is the inventor of the Hydraulic Sinus Condensing (HSC) Technique, widely regarded as a gold standard for minimally invasive sinus elevation. In the mid-1990s, he pioneered the use of hydraulic pressure to access the sinus safely without membrane perforation—an innovation that laid the foundation for SinusFuse.
Dr. Chen has also introduced numerous landmark techniques, including flapless sinus lifts, immediate loading protocols, vertical and lingual augmentation methods, and AI-driven implant planning software. He holds over 50 international patents and trademarks and has received multiple innovation awards, including gold medals at the INPEX Medical Innovation Conference.
As co-founder of the Dental Implant Institute in Las Vegas, Dr. Chen continues to educate clinicians worldwide while shaping the future of precision, nano-invasive implant dentistry.
Conclusion: A New Standard in Sinus Implantology
SinusFuse is more than an implant—it is a comprehensive clinical solution. By integrating sinus elevation, bone management, and implant placement into a single procedure, it reduces surgical complexity, shortens treatment time, and improves patient comfort.
As the world’s first sinus implant, SinusFuse sets a new benchmark for innovation in dental implantology, reflecting a future where advanced engineering and minimally invasive dentistry work seamlessly together to achieve superior clinical outcomes.
Frequently Asked Questions (FAQ) About SinusFuse
1. What makes SinusFuse different from conventional dental implants?
SinusFuse is the world’s first implant specifically engineered to function as a sinus implant. Unlike conventional implants, it integrates multiple surgical steps into one procedure by controlled up-fracturing of the sinus floor, transporting autogenous bone, gently elevating the sinus membrane, and placing the implant simultaneously. This reduces the need for separate sinus lift surgeries.
2. In which clinical situations is SinusFuse most commonly indicated?
SinusFuse is primarily indicated for posterior maxillary cases with limited residual bone height where traditional implants would require sinus augmentation. It is suitable for partially or fully edentulous patients who meet the minimum bone requirements and are candidates for delayed loading protocols.
3. Does SinusFuse eliminate the need for bone grafting materials?
In many cases, yes. SinusFuse is designed to harvest and reposition the patient’s own autogenous bone during placement, which can significantly reduce or eliminate the need for additional grafting materials. However, final clinical decisions depend on individual anatomy and bone quality.
4. Is SinusFuse compatible with standard prosthetic restorations?
Yes. SinusFuse is part of the OsseoFuse Dental Implant System and is compatible with a wide range of prosthetic options, including cement-retained, screw-retained, overdenture, and fixed bridge restorations, provided proper treatment planning is performed.
5. What level of training is required to use SinusFuse safely?
SinusFuse should only be used by clinicians who have received formal training in implant dentistry. Due to its advanced design and specialized surgical protocol, additional education in minimally invasive sinus techniques is strongly recommended to ensure predictable outcomes and minimize complications.
6. What are the main risks associated with SinusFuse implantation?
As with any implant system, potential risks include lack of osseointegration, infection, excessive bone loss, sinus membrane perforation, or implant failure. Proper patient selection, precise surgical technique, and adherence to clinical protocols significantly reduce these risks.
7. How much height can be predictably obtained?
Crestal sinus lift ranges from ~3–5 mm under ideal conditions.
8. What is the minimum horizontal width and vertical height from the alveolar crest to the sinus to securely lift the membrane?
A minimum residual bone height (RBH) of ~2 mm above the sinus floor. Minimum horizontal width >4mm.
Recent Articles
Dental implantology has entered a new era with the introduction of SinusFuse, a groundbreaking innovation recognized as the world’s first true sinus-lifting implant. Designed to address one of the most complex challenges in implant dentistry, insufficient bone height in the posterior maxilla, SinusFuse represents a major technological leap, combining advanced biomechanics, minimally invasive principles, and patented engineering into a single implant system.
Developed under the OsseoFuse Dental Implant System, SinusFuse is not merely an incremental improvement. It is a paradigm shift that redefines how clinicians approach sinus elevation, bone management, and implant placement in compromised maxillary cases.
The Clinical Challenge of Maxillary Sinus Implants
Posterior maxillary implant placement is often complicated by pneumatization of the maxillary sinus and reduced residual bone height following tooth loss. Traditionally, these cases require additional surgical interventions such as lateral window sinus lifts or crestal sinus elevation with grafting materials. While effective, these procedures increase treatment time, cost, patient morbidity, and clinical risk.
SinusFuse was engineered specifically to overcome these limitations by integrating sinus elevation and implant placement into a single, controlled, minimally invasive procedure.
What Is SinusFuse?
SinusFuse is a patented sinus implant within the OsseoFuse Dental Implant System. It is manufactured from high-grade biocompatible titanium and titanium alloys, ensuring excellent osseointegration, mechanical strength, and long-term stability.
Unlike conventional implants, SinusFuse is designed to interact dynamically with the sinus floor. During placement, it performs multiple critical functions simultaneously:
Controlled up-fracture of the sinus floor
Harvesting and transporting autogenous bone
Gentle elevation of the Schneiderian membrane
Immediate implant placement in a single surgical sequence
This integrated functionality eliminates the need for separate sinus lift instruments or grafting procedures in many clinical scenarios.
OsseoFuse Dental Implant System Overview
The OsseoFuse system includes HexaPlus™, ConicalPlus™, and Sinus™ implants, along with compatible abutments, overdenture bars, and restorative components. The system is indicated for use in both the maxilla and mandible, supporting:
Single-tooth restorations
Multiple-unit prostheses
Cement-retained and screw-retained restorations
Overdentures
Fixed bridgework (terminal or intermediate abutments)
The system supports one-stage and two-stage surgical protocols and is intended primarily for delayed loading, allowing predictable osseointegration in a wide range of bone qualities.
Indications for SinusFuse and OsseoFuse Implants
SinusFuse and other OsseoFuse implants are indicated for patients who are partially or fully edentulous, provided adequate bone conditions exist. Minimum bone requirements include:
At least 1 mm of circumferential bone
At least 2 mm of apical bone
They are suitable for cases where traditional implant placement would otherwise require sinus augmentation procedures.
Surgical Planning and Directions for Use
Successful outcomes with SinusFuse rely heavily on proper diagnosis, planning, and surgical execution. Before implant placement, clinicians must evaluate:
Vertical dimension and restorative space
Distance between the alveolar crest and opposing dentition
Abutment selection and final prosthetic design
Importantly, the final prosthesis should be planned before implant placement to ensure restorative-driven implant positioning.
During surgery, continuous sterile irrigation is mandatory to prevent thermal trauma and protect surrounding tissues. Excessive drilling pressure must be avoided, and only sharp, high-quality instruments should be used to preserve bone vitality and enhance osseointegration.
Contraindications and Patient Selection
SinusFuse implants should not be placed in patients with:
Severely insufficient bone volume or density
Poor oral hygiene
Heavy tobacco use
Uncontrolled systemic conditions such as diabetes or blood disorders
Inadequate patient selection or compromised bone conditions significantly increase the risk of implant failure or lack of osseointegration.
Warnings and Precautions
Dental implant surgery is a highly specialized clinical procedure. Proper training in implantology is strongly recommended before using the OsseoFuse system. Incorrect technique may lead to implant instability, excessive bone loss, or restoration failure.
Additional precautions include:
Avoiding electrosurgical devices or lasers near metallic implants
Monitoring for signs of implant mobility, infection, or bone loss
Immediate intervention or removal if implant failure is suspected
Preventing aspiration or swallowing of small components
Avoiding small-diameter implants with angled abutments in posterior regions
The OsseoFuse system has not been evaluated for MRI safety, and compatibility in MR environments has not been established.
Sterility, Cleaning, and Instrument Handling
OsseoFuse implant fixtures, mounts, and cover screws are supplied sterile via gamma irradiation and are ready for use. Abutments, accessories, and surgical instruments are provided non-sterile and must be sterilized before use.
Recommended cleaning protocol includes:
Ultrasonic cleaning for 10 minutes
Manual removal of debris with a soft-bristle brush
Thorough rinsing, including an alcohol rinse to prevent corrosion
Complete air drying
Steam sterilization using validated cycles
These sterilization parameters are validated to a Sterility Assurance Level (SAL) of 10⁻⁶ in accordance with FDA-recognized ISO standards.
Healing, Restoration, and Occlusal Considerations
Healing time varies based on bone quality, tissue response, and surgical assessment. During this period, excessive functional loading must be avoided. Proper occlusal design is essential to prevent overload, which can compromise osseointegration and long-term implant success.
Potential Adverse Events
As with any implant system, potential complications may include:
Failure or loss of osseointegration
Bone dehiscence requiring grafting
Sinus or anatomical structure perforation
Infection, abscess, or inflammation
Persistent pain or altered sensation
Excessive bone loss
Implant fracture
Early diagnosis and intervention are critical to managing these risks effectively.

The Innovator Behind SinusFuse: Dr. C. Leon Chen
Meet the Innovator Behind SinusFuse: Dr. C. Leon Chen
The vision behind SinusFuse belongs to Dr. C. Leon Chen, DMD, MS, DBA, a globally respected authority in implant dentistry and minimally invasive techniques. A graduate of Harvard University School of Dental Medicine and Northwestern University’s Department of Periodontics, Dr. Chen has dedicated over three decades to advancing implant science.
He is the inventor of the Hydraulic Sinus Condensing (HSC) Technique, widely regarded as a gold standard for minimally invasive sinus elevation. In the mid-1990s, he pioneered the use of hydraulic pressure to access the sinus safely without membrane perforation—an innovation that laid the foundation for SinusFuse.
Dr. Chen has also introduced numerous landmark techniques, including flapless sinus lifts, immediate loading protocols, vertical and lingual augmentation methods, and AI-driven implant planning software. He holds over 50 international patents and trademarks and has received multiple innovation awards, including gold medals at the INPEX Medical Innovation Conference.
As co-founder of the Dental Implant Institute in Las Vegas, Dr. Chen continues to educate clinicians worldwide while shaping the future of precision, nano-invasive implant dentistry.
Conclusion: A New Standard in Sinus Implantology
SinusFuse is more than an implant—it is a comprehensive clinical solution. By integrating sinus elevation, bone management, and implant placement into a single procedure, it reduces surgical complexity, shortens treatment time, and improves patient comfort.
As the world’s first sinus implant, SinusFuse sets a new benchmark for innovation in dental implantology, reflecting a future where advanced engineering and minimally invasive dentistry work seamlessly together to achieve superior clinical outcomes.
Frequently Asked Questions (FAQ) About SinusFuse
1. What makes SinusFuse different from conventional dental implants?
SinusFuse is the world’s first implant specifically engineered to function as a sinus implant. Unlike conventional implants, it integrates multiple surgical steps into one procedure by controlled up-fracturing of the sinus floor, transporting autogenous bone, gently elevating the sinus membrane, and placing the implant simultaneously. This reduces the need for separate sinus lift surgeries.
2. In which clinical situations is SinusFuse most commonly indicated?
SinusFuse is primarily indicated for posterior maxillary cases with limited residual bone height where traditional implants would require sinus augmentation. It is suitable for partially or fully edentulous patients who meet the minimum bone requirements and are candidates for delayed loading protocols.
3. Does SinusFuse eliminate the need for bone grafting materials?
In many cases, yes. SinusFuse is designed to harvest and reposition the patient’s own autogenous bone during placement, which can significantly reduce or eliminate the need for additional grafting materials. However, final clinical decisions depend on individual anatomy and bone quality.
4. Is SinusFuse compatible with standard prosthetic restorations?
Yes. SinusFuse is part of the OsseoFuse Dental Implant System and is compatible with a wide range of prosthetic options, including cement-retained, screw-retained, overdenture, and fixed bridge restorations, provided proper treatment planning is performed.
5. What level of training is required to use SinusFuse safely?
SinusFuse should only be used by clinicians with formal training in implant dentistry. Due to its advanced design and specialized surgical protocol, additional education in minimally invasive sinus techniques is strongly recommended to ensure predictable outcomes and minimize complications.
6. What are the main risks associated with SinusFuse implantation?
As with any implant system, potential risks include lack of osseointegration, infection, excessive bone loss, sinus membrane perforation, or implant failure. Proper patient selection, precise surgical technique, and adherence to clinical protocols significantly reduce these risks.
7. How much height can be predictably obtained?
Crestal sinus lift ranges from ~3–5 mm under ideal conditions.
8. What is the minimum horizontal width and vertical height from the alveolar crest to the sinus to securely lift the membrane?
A minimum residual bone height (RBH) of ~2 mm above the sinus floor. Minimum horizontal width >4mm.
Recent Articles
Peri-Implantitis Treatment & Prevention Methods
Peri-Implantitis Treatment & Prevention Methods Peri-implantitis remains one of the most significant biological complications affecting dental implants, posing a serious threat to long-term implant [...]
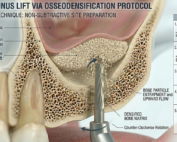
Latest Sinus Lifting Techniques in Modern Implant Dentistry
Latest Sinus Lifting Techniques in Modern Implant Dentistry A Comprehensive Clinical Review Introduction to Sinus Lifting in Implantology Sinus lifting, or maxillary sinus floor [...]
Biomechanics of the Triple Abutment & BOPiT Concept
Dental Biomechanics · Implant Science · Clinical Evidence Biomechanics of the Triple Abutment & BOPiT Concept How a saddle-shaped mathematical surface is rewriting the rules of load distribution [...]
The Woman Who Proved One Implant Could Hold Three Crowns
The Woman Who Proved One Implant Could Hold Three Crowns While the dental establishment looked away, Dr. Luciana Colepícolo spent 12 years building the [...]
ENPP1, The Molecular Brake That Limits Tissue Healing
ENPP1, The Molecular Brake That Limits Tissue Healing Introduction: A New Biological Barrier to Dental Regeneration Meet ENPP1—a protein most dentists have never heard [...]
The Rise of Self‑Healing Dental Biomaterials
The Rise of Self‑Healing Dental Biomaterials Introduction In modern restorative dentistry, durability and longevity of materials remain among the greatest clinical challenges. Traditional dental [...]