A Promising Natural Powder from Brazil: Morin for Gingivitis
A Promising Natural Powder from Brazil: Morin for Gingivitis
Morin Powder: Natural Brazilian Remedy for Gum Disease Care
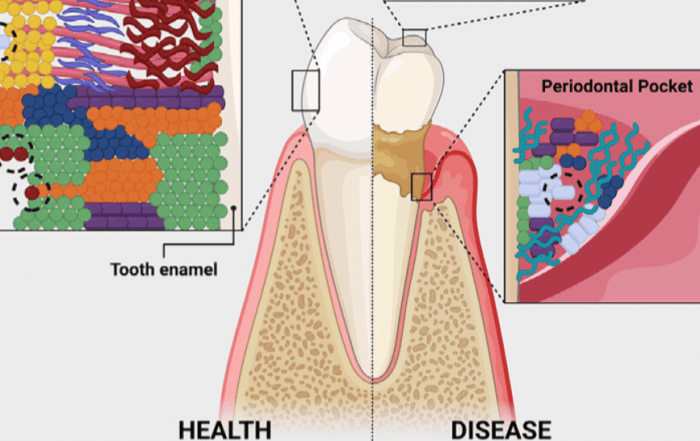
Researchers from São Paulo State University (UNESP), Brazil, have made a significant advance in periodontal (gum) disease treatment by introducing morin powder. They have developed a natural, plant-derived controlled-release morin powder that shows strong antimicrobial, anti-inflammatory, antioxidant, and anti-biofilm effects. This powder, when released slowly via polymer systems, may serve as a promising alternative to traditional antibiotics. The detailed findings were published in Archives of Oral Biology (DOI: 10.1016/j.archoralbio.2025.106343). (1)
Below is a deep dive into what the study reveals, how the morin powder works, scientific context, potential applications, limitations, and what future research is needed.
What Is Morin Powder? A Flavonoid from Plants
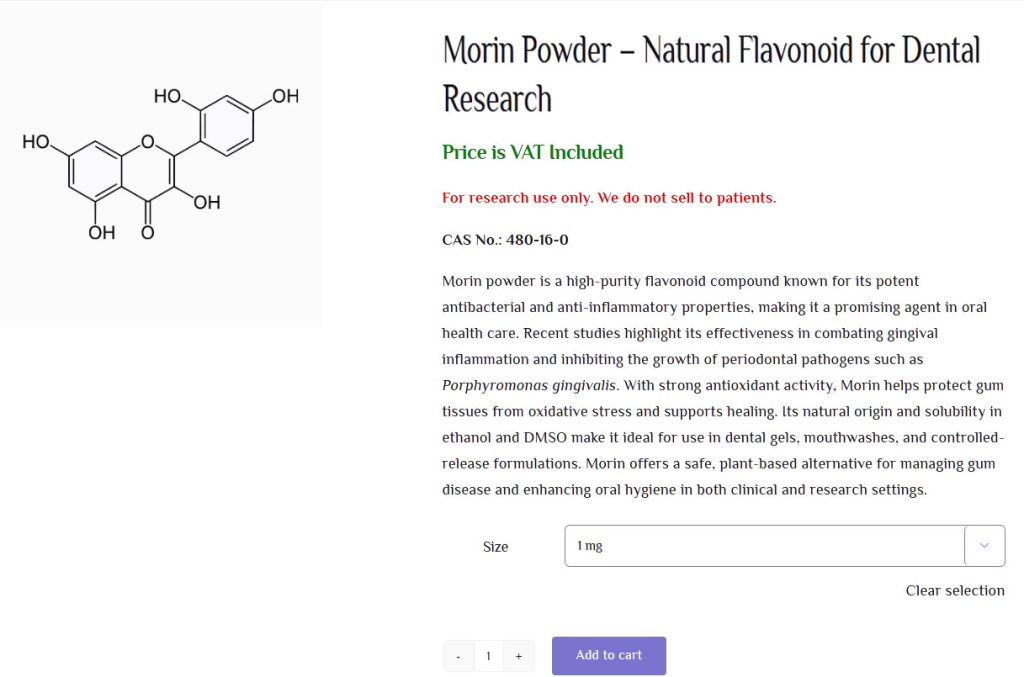
Chemical nature: Morin powder is a flavonoid—a type of natural compound found in many plants; known for its antioxidant, anti-inflammatory, and antimicrobial properties. (1)
Sources: It occurs in guava leaves, apple peels, fig skins, certain teas, and almonds. But consuming these foods alone generally does not provide enough morin to achieve medicinal benefit, especially for oral health. (2)
The São Paulo Study: What the Researchers Did
The study, led by Luciana Solera Sales (during her PhD under Fernanda Lourenção Brighenti), involved experiments in vitro (in the lab) using epithelial cell cultures, neutrophils, and multispecies bacterial biofilms to simulate the conditions in human mouths affected by periodontal disease. (2)
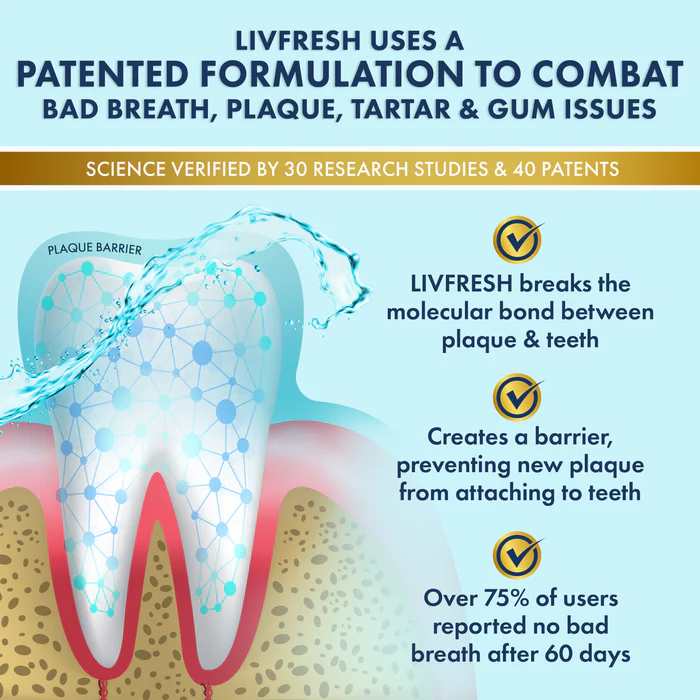
Formulation: Morin was processed into a fine powder via spray drying. This technique is similar to how powdered milk is made. The powder is then embedded in polymers (such as gellan gum or sodium alginate) to allow controlled release; that is, to allow morin to stay in the mouth environment (on the gums, teeth, mucous membrane) for a longer time, resisting wash-away by saliva. (1)
Testing:
Antimicrobial / anti-biofilm: Morin was tested against several bacterial pathogens known to contribute to gum disease (e.g., Fusobacterium nucleatum, Porphyromonas gingivalis) and in a multispecies biofilm model. The goal was to assess both planktonic (free-floating) bacteria and biofilm (bacteria embedded in a matrix) survival. (1)
Inflammatory response: Epithelial cells (H400 line) were pre-treated with morin (free or in formulation), then exposed to heat-killed bacterial pathogens. The study measured production of pro-inflammatory markers (GM-CSF), gene expression of IL-8, IL1B, NF-κB, NLRP3, etc. (1)
Oxidative stress/antioxidant: Also assessed was the production of reactive oxygen species by neutrophils challenged with bacteria, with or without morin. (1)
Cytotoxicity: Researchers ensured that morin (both free and released from polymers) did not excessively harm human oral keratinocytes (cells lining the mouth) except potentially at very high doses. The safe doses were identified. (1)
Key Results: How Effective Was the Morin Powder?
Here are the main outcomes:
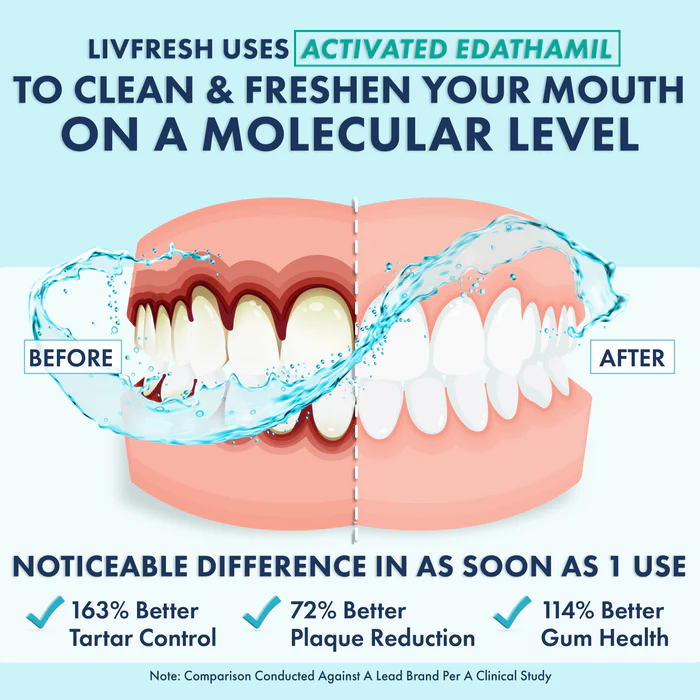
Reduced bacterial viability & biofilm biomass: Morin (free or in slow-release polymer systems) significantly lowered the number of bacteria and reduced biofilm biomass in multi-species biofilms. (1)
Lowered inflammation: Levels of GM-CSF secretion were reduced, gene expression of IL-8, IL1B, NF-κB, and LRP3 were also significantly down. These are markers associated with inflammation. (1)
Antioxidant effect: The morin treatment reduced reactive oxygen species (ROS) production in neutrophils challenged with periodontal pathogens. (1)
Control of acidity and extracellular matrix: In the films/tablets with polymer systems, morin reduced acid production in biofilms, as well as dry weight and insoluble extracellular polysaccharides (these polysaccharides help bacteria stick together and form the biofilm matrix). (1)
Minimal cytotoxicity at effective doses: Only at the highest concentrations was there noticeable cytotoxicity; lower, therapeutically relevant doses were well tolerated by oral cells. (1)
Advantages of Morin Powder Over Existing Treatments
Alternative to antibiotics: Because of morin’s antimicrobial effects on plaque-forming bacteria, it may serve as a substitute or adjunct to antibiotics, reducing risks of antibiotic resistance. (2)
Fewer side effects: Traditional chemical antimicrobial or antiseptic agents often lead to mouth irritation, altered taste, teeth staining, or imbalance of the oral microbiome. The morin powder, due to its natural origin and controlled release, seems less likely to cause such adverse outcomes. (2)
Suitability for people with limited dental care ability: The powder format, particularly in slow-release formulations like films, tablets, or polymers that adhere to mucosa, has potential for populations who have difficulty brushing or flossing properly (elderly, disabled). (2)
Challenges and Technical Considerations of Morin Powder
Even promising natural compounds come with hurdles; the study and related research point out several:
Solubility and Stability: Morin is not very water-soluble, and in the mouth environment, substances get quickly washed away by saliva. Therefore, creating polymer-based, mucoadhesive systems is essential for maintaining effective concentration at the site of action. (2)
Controlled Release Design: The right polymer(s) must be chosen to release morin over time in a manner that’s effective but safe; release that’s too fast loses benefit, too slow might not reach needed levels. Gellan gum and sodium alginate are among the polymers tested.
Cytotoxicity & Dosage Control: It’s critical to ensure that beneficial antimicrobial/inflammatory-modulating doses do not damage healthy oral tissues. There is evidence that high doses can be cytotoxic, so safety margins need to be established.
Scaling up production & regulatory approval: Translating lab results into commercial oral care products (toothpaste, mouthwash, lozenges, etc.) requires proving safety & efficacy in animal then human trials, quality control in manufacturing, and obtaining regulatory approvals.
Scientific Context of Morin Powder: Related Studies & Mechanisms
“Anti-inflammatory, antioxidant, and antimicrobial evaluation of morin” (Archives of Oral Biology) is the core recent paper for this morin powder approach. It provides detailed in vitro evidence of the effects summarized above. (3)
Another related study: Mucoadhesive controlled-release formulations containing morin (UNESP et al.) evaluated morin in films or tablets based on gellan gum, testing with polymicrobial biofilms derived from saliva, exposed to sucrose, etc. They looked at viability, acidogenicity, extracellular polysaccharides, dry weight, and cytotoxicity in keratinocytes. (3)
Broader reviews of natural bioactive compounds in periodontal disease place morin among many flavonoids and polyphenols that act by:
Disrupting biofilms (blocking adhesion, interfering with extracellular polymeric substances)
Modulating immune/inflammatory pathways (NF-κB, NLRP3 inflammasome, IL-1β, IL-6, TNF-α, etc.)
Neutralizing oxidative stress (reducing ROS)
Maintaining or restoring microbial balance, i.e., suppressing pathogens without eliminating beneficial oral bacteria. (3)
Potential Products & Applications of Morin Powder
Based on the study, possible future commercial or clinical products include:
Toothpaste or gels infused with morin in controlled-release polymers.
Mouthwashes or rinses that deposit morin-containing films or micro-particles on gums and mucosa.
Slow-release lozenges or tablets that dissolve gradually, maintaining anti-microbial contact in the mouth.
Films or patches (mucoadhesive) that can be applied to gums directly, especially useful in periodontal pockets during non-surgical therapy.
These could be particularly useful for:
Elderly individuals or people with physical or motor impairments who cannot maintain rigorous tooth-brushing.
Patients suffering from chronic gum inflammation or periodontitis, for whom antibiotics are less desirable or have side effects.
Preventive care, to reduce the progression of gingivitis to periodontitis.
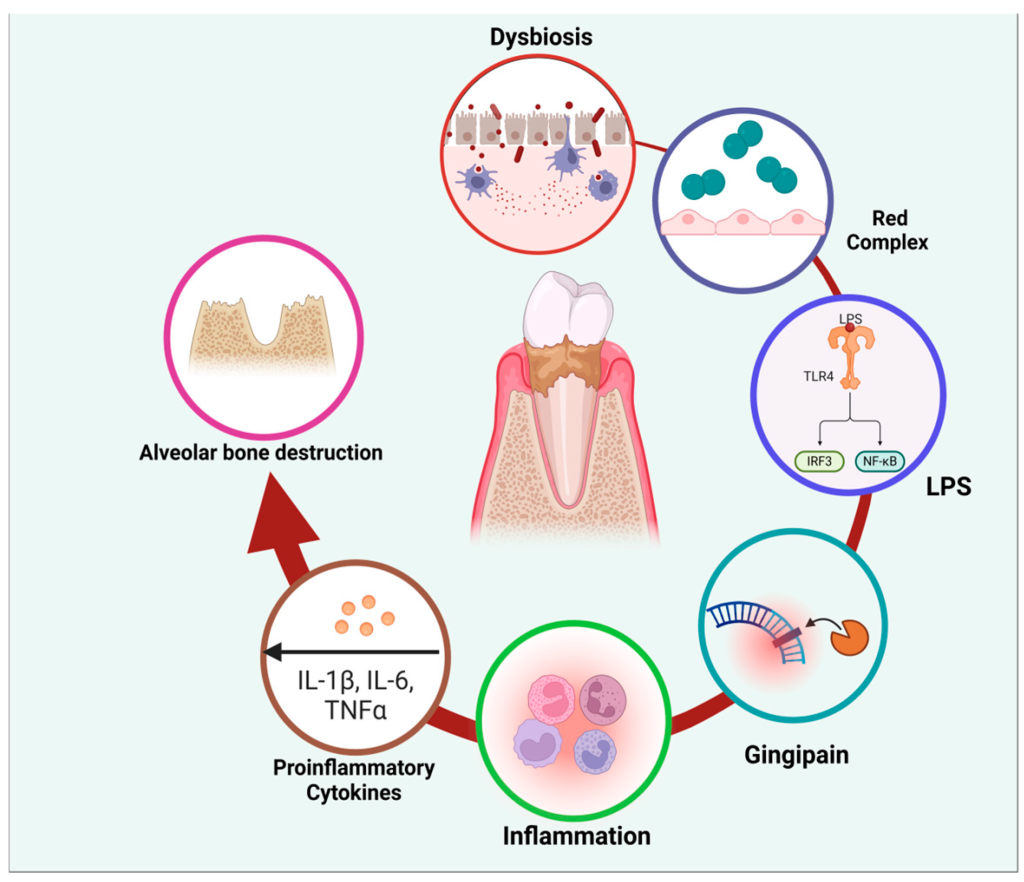
Pathophysiological cycle of periodontitis
Figure 1. Illustrates the pathophysiological cycle of periodontitis, beginning with dysbiosis, where microbial imbalance initiates disease progression. The formation of biofilm and colonization by red-complex bacteria exacerbate inflammation through the release of lipopolysaccharides (LPS). This stimulates pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α, leading to gingival pain and progressive alveolar bone destruction. The inflammatory response further sustains dysbiosis, creating a vicious cycle that perpetuates tissue degradation and disease advancement.
Limitations and Future Research of Morin Powder
While the lab results are promising, there are important limitations and steps ahead:
In vitro vs. in vivo: All current studies have been conducted in laboratory settings (cells, biofilms). Real mouths are more complex: saliva flow, varied microbiomes, and immune responses. Human clinical trials are needed.
Dose and safety in humans: The window between effective dose and toxicity must be fully mapped in animals and then in human trials.
Long-term effects: Effects of continuous or repeated use of morin-containing products need evaluation: stability, accumulation, potential for resistance, and effects on beneficial bacteria.
Delivery system optimization: Polymers used must be safe, biocompatible, taste acceptable, stable in products, and able to release morin at effective concentrations over meaningful time periods.
Regulatory, manufacturing, cost: For any morin-based product to reach consumers, it must meet regulatory standards (e.g. for cosmetics, drugs, oral hygiene products), be producible at scale, be stable in storage, and be cost-effective.
Implications of Morin Powder for Oral Health
Reduced reliance on antibiotics: Widespread use of antibiotics in dentistry contributes to antibiotic resistance. Natural antimicrobial agents like morin might help limit the use of systemic antibiotics or reduce the need for strong local antiseptics.
Accessible preventive care: For communities or populations with limited access to dental care, or for those with special abilities, products with morin that need less mechanical effort (e.g., slow-release patches) could help maintain gum health.
Lower cost natural options: Because morin comes from widely available plant sources, and if manufacturing is optimized, morin-based products could be cheaper than some synthetic antimicrobials.
Enrollment in broader natural bioactive compound strategies: As reviews suggest, compounds like morin may be most effective when used in combination with proper hygiene (brushing, flossing), professional cleaning, and possibly diet / antioxidant support.
Conclusion
The recent studies from São Paulo provide strong evidence that morin powder, especially when formulated in controlled-release polymer systems, holds real promise as an alternative or adjunct in the treatment and prevention of gum disease. It addresses microbial biofilms, inflammation, and oxidative stress—all key players in periodontitis.
However, lab successes are just the first step. Clinical trials in animals and humans, safety and dosage studies, product formulation and acceptability, regulatory approval, and long-term monitoring will be necessary before Morin powder products can become commercially available and widely used.
Still, for anyone interested in natural oral health solutions, gum disease prevention, or antimicrobial agents without heavy side effects, morin looks like one of the most promising flavonoids of recent years.
References
1- New Insights in Natural Bioactive Compounds for Periodontal Disease: Advanced Molecular Mechanisms and Therapeutic Potential
2- Plant-derived compound has antimicrobial and anti-inflammatory effects against periodontal disease
3- Anti-inflammatory, antioxidant, and antimicrobial evaluation of morin
Recent Articles
Researchers from São Paulo State University (UNESP), Brazil, have made a significant advance in periodontal (gum) disease treatment by introducing morin powder. They have developed a natural, plant-derived controlled-release morin powder that shows strong antimicrobial, anti-inflammatory, antioxidant, and anti-biofilm effects. This powder, when released slowly via polymer systems, may serve as a promising alternative to traditional antibiotics. The detailed findings were published in Archives of Oral Biology (DOI: 10.1016/j.archoralbio.2025.106343). (1)
Below is a deep dive into what the study reveals, how the morin powder works, scientific context, potential applications, limitations, and what future research is needed.
What Is Morin Powder? A Flavonoid from Plants
Chemical nature: Morin powder is a flavonoid—a type of natural compound found in many plants; known for its antioxidant, anti-inflammatory, and antimicrobial properties. (1)
Sources: It occurs in guava leaves, apple peels, fig skins, certain teas, and almonds. But consuming these foods alone generally does not provide enough morin to achieve medicinal benefit, especially for oral health. (2)
The São Paulo Study: What the Researchers Did
The study, led by Luciana Solera Sales (during her PhD under Fernanda Lourenção Brighenti), involved experiments in vitro (in the lab) using epithelial cell cultures, neutrophils, and multispecies bacterial biofilms to simulate the conditions in human mouths affected by periodontal disease. (2)
Formulation: Morin was processed into a fine powder via spray drying. This technique is similar to how powdered milk is made. The powder is then embedded in polymers (such as gellan gum or sodium alginate) to allow controlled release; that is, to allow morin to stay in the mouth environment (on the gums, teeth, mucous membrane) for a longer time, resisting wash-away by saliva. (1)
Testing:
Antimicrobial / anti-biofilm: Morin was tested against several bacterial pathogens known to contribute to gum disease (e.g., Fusobacterium nucleatum, Porphyromonas gingivalis) and in a multispecies biofilm model. The goal was to assess both planktonic (free-floating) bacteria and biofilm (bacteria embedded in a matrix) survival. (1)
Inflammatory response: Epithelial cells (H400 line) were pre-treated with morin (free or in formulation), then exposed to heat-killed bacterial pathogens. The study measured production of pro-inflammatory markers (GM-CSF), gene expression of IL-8, IL1B, NF-κB, NLRP3, etc. (1)
Oxidative stress/antioxidant: Also assessed was the production of reactive oxygen species by neutrophils challenged with bacteria, with or without morin. (1)
Cytotoxicity: Researchers ensured that morin (both free and released from polymers) did not excessively harm human oral keratinocytes (cells lining the mouth) except potentially at very high doses. The safe doses were identified. (1)
Key Results: How Effective Was the Morin Powder?
Here are the main outcomes:
Reduced bacterial viability & biofilm biomass: Morin (free or in slow-release polymer systems) significantly lowered the number of bacteria and reduced biofilm biomass in multi-species biofilms. (1)
Lowered inflammation: Levels of GM-CSF secretion were reduced, gene expression of IL-8, IL1B, NF-κB, and LRP3 were also significantly down. These are markers associated with inflammation. (1)
Antioxidant effect: The morin treatment reduced reactive oxygen species (ROS) production in neutrophils challenged with periodontal pathogens. (1)
Control of acidity and extracellular matrix: In the films/tablets with polymer systems, morin reduced acid production in biofilms, as well as dry weight and insoluble extracellular polysaccharides (these polysaccharides help bacteria stick together and form the biofilm matrix). (1)
Minimal cytotoxicity at effective doses: Only at the highest concentrations was there noticeable cytotoxicity; lower, therapeutically relevant doses were well tolerated by oral cells. (1)
Advantages of Morin Powder Over Existing Treatments
Alternative to antibiotics: Because of morin’s antimicrobial effects on plaque-forming bacteria, it may serve as a substitute or adjunct to antibiotics, reducing risks of antibiotic resistance. (2)
Fewer side effects: Traditional chemical antimicrobial or antiseptic agents often lead to mouth irritation, altered taste, teeth staining, or imbalance of the oral microbiome. The morin powder, due to its natural origin and controlled release, seems less likely to cause such adverse outcomes. (2)
Suitability for people with limited dental care ability: The powder format, particularly in slow-release formulations like films, tablets, or polymers that adhere to mucosa, has potential for populations who have difficulty brushing or flossing properly (elderly, disabled). (2)
Challenges and Technical Considerations of Morin Powder
Even promising natural compounds come with hurdles; the study and related research point out several:
Solubility and Stability: Morin is not very water-soluble, and in the mouth environment, substances get quickly washed away by saliva. Therefore, creating polymer-based, mucoadhesive systems is essential for maintaining effective concentration at the site of action. (2)
Controlled Release Design: The right polymer(s) must be chosen to release morin over time in a manner that’s effective but safe; release that’s too fast loses benefit, too slow might not reach needed levels. Gellan gum and sodium alginate are among the polymers tested.
Cytotoxicity & Dosage Control: It’s critical to ensure that beneficial antimicrobial/inflammatory-modulating doses do not damage healthy oral tissues. There is evidence that high doses can be cytotoxic, so safety margins need to be established.
Scaling up production & regulatory approval: Translating lab results into commercial oral care products (toothpaste, mouthwash, lozenges, etc.) requires proving safety & efficacy in animal then human trials, quality control in manufacturing, and obtaining regulatory approvals.
Scientific Context of Morin Powder: Related Studies & Mechanisms
“Anti-inflammatory, antioxidant, and antimicrobial evaluation of morin” (Archives of Oral Biology) is the core recent paper for this morin powder approach. It provides detailed in vitro evidence of the effects summarized above. (3)
Another related study: Mucoadhesive controlled-release formulations containing morin (UNESP et al.) evaluated morin in films or tablets based on gellan gum, testing with polymicrobial biofilms derived from saliva, exposed to sucrose, etc. They looked at viability, acidogenicity, extracellular polysaccharides, dry weight, and cytotoxicity in keratinocytes. (3)
Broader reviews of natural bioactive compounds in periodontal disease place morin among many flavonoids and polyphenols that act by:
Disrupting biofilms (blocking adhesion, interfering with extracellular polymeric substances)
Modulating immune/inflammatory pathways (NF-κB, NLRP3 inflammasome, IL-1β, IL-6, TNF-α, etc.)
Neutralizing oxidative stress (reducing ROS)
Maintaining or restoring microbial balance, i.e., suppressing pathogens without eliminating beneficial oral bacteria. (3)
Potential Products & Applications of Morin Powder
Based on the study, possible future commercial or clinical products include:
Toothpaste or gels infused with morin in controlled-release polymers.
Mouthwashes or rinses that deposit morin-containing films or micro-particles on gums and mucosa.
Slow-release lozenges or tablets that dissolve gradually, maintaining anti-microbial contact in the mouth.
Films or patches (mucoadhesive) that can be applied to gums directly, especially useful in periodontal pockets during non-surgical therapy.
These could be particularly useful for:
Elderly individuals or people with physical or motor impairments who cannot maintain rigorous tooth-brushing.
Patients suffering from chronic gum inflammation or periodontitis, for whom antibiotics are less desirable or have side effects.
Preventive care, to reduce the progression of gingivitis to periodontitis.

Pathophysiological cycle of periodontitis
Figure 1. Illustrates the pathophysiological cycle of periodontitis, beginning with dysbiosis, where microbial imbalance initiates disease progression. The formation of biofilm and colonization by red-complex bacteria exacerbate inflammation through the release of lipopolysaccharides (LPS). This stimulates pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α, leading to gingival pain and progressive alveolar bone destruction. The inflammatory response further sustains dysbiosis, creating a vicious cycle that perpetuates tissue degradation and disease advancement.
Limitations and Future Research of Morin Powder
While the lab results are promising, there are important limitations and steps ahead:
In vitro vs. in vivo: All current studies have been conducted in laboratory settings (cells, biofilms). Real mouths are more complex: saliva flow, varied microbiomes, and immune responses. Human clinical trials are needed.
Dose and safety in humans: The window between effective dose and toxicity must be fully mapped in animals and then in human trials.
Long-term effects: Effects of continuous or repeated use of morin-containing products need evaluation: stability, accumulation, potential for resistance, and effects on beneficial bacteria.
Delivery system optimization: Polymers used must be safe, biocompatible, taste acceptable, stable in products, and able to release morin at effective concentrations over meaningful time periods.
Regulatory, manufacturing, cost: For any morin-based product to reach consumers, it must meet regulatory standards (e.g. for cosmetics, drugs, oral hygiene products), be producible at scale, be stable in storage, and be cost-effective.
Implications of Morin Powder for Oral Health
Reduced reliance on antibiotics: Widespread use of antibiotics in dentistry contributes to antibiotic resistance. Natural antimicrobial agents like morin might help limit the use of systemic antibiotics or reduce the need for strong local antiseptics.
Accessible preventive care: For communities or populations with limited access to dental care, or for those with special abilities, products with morin that need less mechanical effort (e.g., slow-release patches) could help maintain gum health.
Lower cost natural options: Because morin comes from widely available plant sources, and if manufacturing is optimized, morin-based products could be cheaper than some synthetic antimicrobials.
Enrollment in broader natural bioactive compound strategies: As reviews suggest, compounds like morin may be most effective when used in combination with proper hygiene (brushing, flossing), professional cleaning, and possibly diet / antioxidant support.
Conclusion
The recent studies from São Paulo provide strong evidence that morin powder, especially when formulated in controlled-release polymer systems, holds real promise as an alternative or adjunct in the treatment and prevention of gum disease. It addresses microbial biofilms, inflammation, and oxidative stress—all key players in periodontitis.
However, lab successes are just the first step. Clinical trials in animals and humans, safety and dosage studies, product formulation and acceptability, regulatory approval, and long-term monitoring will be necessary before Morin powder products can become commercially available and widely used.
Still, for anyone interested in natural oral health solutions, gum disease prevention, or antimicrobial agents without heavy side effects, morin looks like one of the most promising flavonoids of recent years.
References
1- New Insights in Natural Bioactive Compounds for Periodontal Disease: Advanced Molecular Mechanisms and Therapeutic Potential
2- Plant-derived compound has antimicrobial and anti-inflammatory effects against periodontal disease
3- Anti-inflammatory, antioxidant, and antimicrobial evaluation of morin
Recent Articles
Effect of Dental Implant Overdentures on Cognitive Functions
Effect of Dental Implant Overdentures on Cognitive Functions Recent research increasingly highlights the link between oral health and brain function, suggesting that tooth loss [...]
A New Link Between Periodontal Disease and Breast Cancer
A New Link Discovered Between Periodontal Disease and Breast Cancer Emerging evidence is redefining how oral health intersects with systemic disease. A growing body [...]
The Link Between Flossing and Cardiovascular & Brain Health
The Link Between Flossing and Cardiovascular & Brain Health Recent research presented at the American Stroke Association’s 2025 conference highlights a remarkable connection between [...]
Tubarial Glands: The Nasopharynx Hidden Salivary Glands
Tubarial Glands: The Nasopharynx Hidden Salivary Glands Introduction: A Hidden Anatomical Surprise In 2020, scientists made a discovery that shook the foundations of human [...]
Morin Powder: Natural Brazilian Remedy for Gum Disease Care
Morin Powder: Natural Brazilian Remedy for Gum Disease Care Researchers from São Paulo State University (UNESP), Brazil, have made a significant advance in periodontal [...]
Not All Dental Plaque is Evil
Not All Dental Plaque is Evil: Some Bacteria Might Be Your Mouth’s Best Friends! 🧪 What Is Dental Plaque? Dental plaque isn’t just goo [...]