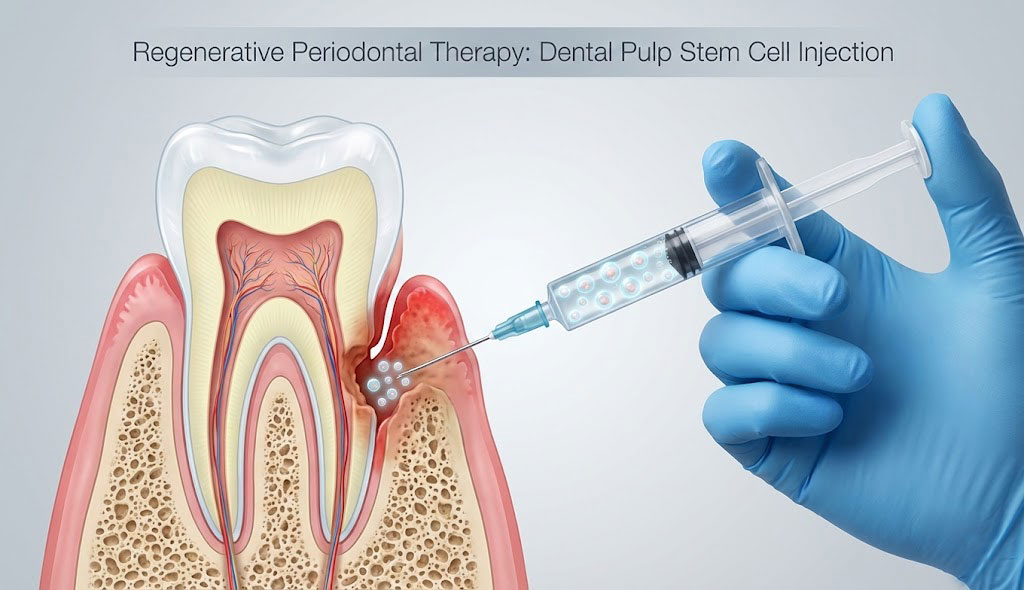
Treating Periodontal Disease with Dental Stem Cell Injection
A Minimally Invasive Breakthrough in Periodontal Regeneration
In our previous article, we examined emerging evidence linking periodontal pathogens to systemic disease, including their potential role in breast cancer progression. That discussion underscored a critical reality: periodontal disease is not merely a localized oral condition, but a chronic inflammatory disease with far-reaching biological implications.
Affecting more than 700 million people worldwide, periodontal disease remains one of the leading causes of tooth loss in adults. It is characterized by progressive destruction of the alveolar bone, periodontal ligament, and connective tissue attachment apparatus. In advanced cases—particularly stage III disease—management of intrabony defects and attachment loss presents a substantial clinical challenge.
Conventional regenerative approaches, such as guided tissue regeneration (GTR), bone grafting, and the use of biologic mediators, including growth factors or enamel matrix derivatives, rely on invasive surgical protocols. While effective in selected cases, these procedures are associated with postoperative morbidity, patient discomfort, infection risk, and inconsistent regenerative outcomes.
In this article, we explore one of the most innovative developments in periodontal therapy based on recent clinical research: dental pulp stem cell (DPSC) injection therapy. This minimally invasive, biologically driven approach is designed to restore periodontal microenvironment homeostasis and stimulate regeneration of both soft and hard tissues—without the need for surgical intervention.
Understanding Periodontal Disease and the Need for Regenerative Innovation
Periodontitis is driven by dysbiotic biofilms and a dysregulated host immune response. The resulting chronic inflammation leads to:
Clinical attachment loss (AL)
Increased periodontal probing depth (PD)
Gingival recession (GR)
Tooth mobility (TM)
Intrabony defects and alveolar bone resorption
While scaling and root planing (SRP) effectively reduces microbial burden, it does not reliably regenerate lost bone or periodontal attachment in moderate-to-severe cases. Surgical regenerative techniques attempt to address this limitation but often yield incomplete or unpredictable tissue reconstruction.
The complexity of periodontal regeneration lies in restoring a coordinated architecture consisting of cementum, periodontal ligament, and alveolar bone. Achieving this requires more than mechanical debridement—it requires biologic modulation.
Why Dental Pulp Stem Cells?
Dental pulp stem cells (DPSCs) are mesenchymal stem cells derived from extracted third molars or orthodontically indicated teeth. Compared with bone marrow–derived or adipose-derived mesenchymal stem cells, DPSCs offer several advantages:
Minimally invasive harvesting
High proliferative capacity
Strong osteogenic differentiation potential
Resistance to inflammation-induced senescence
Low immunogenicity
Immunomodulatory properties
DPSCs express canonical mesenchymal markers CD73, CD90, and CD105, while lacking hematopoietic and immune lineage markers such as CD19, CD34, CD45, CD11b, and HLA-DR. They demonstrate robust osteogenic gene expression, including ALP, RUNX2, BMP2, OPN, and OSX.
Beyond differentiation, DPSCs actively regulate the immune microenvironment. They modulate T cells, B cells, macrophages, dendritic cells, and neutrophils, contributing to the restoration of periodontal microenvironment balance—an essential prerequisite for tissue regeneration.
First-in-Human Randomized Clinical Trials of DPSC Injection
To evaluate safety and efficacy, two randomized, placebo-controlled clinical trials were conducted in China, enrolling 132 patients with chronic periodontitis (158 treated teeth).
Study Design Overview
Total participants: 132
Centers: Two clinical institutions
Primary endpoint: Attachment loss (AL) at 6 months
Secondary endpoints: PD, GR, TM, bone defect depth (BDD)
Intervention: DPSC injection vs saline placebo
Patients received standard periodontal therapy, including oral hygiene instruction and mechanical debridement prior to injection.
In the investigator-initiated trial:
Single injection group: 1 × 10⁷ DPSCs
Double injection group: 1 × 10⁷ DPSCs × 2 (30-day interval)
Saline control group
In the Phase I dose-escalation trial:
Doses ranged from 1 × 10⁶ to 1 × 10⁷ DPSCs per tooth
All injections were delivered directly into the intrabony defect using a minimally invasive technique following SRP.
Safety Profile: Favorable and Well Tolerated
A critical barrier to stem cell therapy adoption is safety. Across both trials:
No serious adverse events were reported
Mild adverse events were limited to grade 1 (toothache, gingival swelling, mild diarrhea)
All adverse effects resolved spontaneously without intervention
Cell preparations demonstrated:
90% viability
Absence of bacterial contamination
Minimal residual processing enzymes
The findings confirm that allogeneic DPSC injection is safe and well-tolerated in the treatment of periodontal disease.

DPSC injection demonstrated greater efficacy in bone defect regeneration compared to saline injection. a, c In narrow initial bone defects, CBCT scans show that DPSC injection significantly reduced the alveolar bone resorption height at day 180 post-treatment. Notably, DPSC injection also reduced signs of periodontitis-induced maxillary sinus inflammation in some cases, as indicated by the yellow dotted line in (c), providing further evidence of its therapeutic effects on local inflammation. b, d In flat initial bone defects, CBCT scans also revealed therapeutic benefits of DPSC injection, although to a lesser extent compared with the narrow defect group. The yellow stars indicate the chief complaint tooth, and the yellow arrows indicate the bone defect
Clinical Efficacy: Regeneration of Both Soft and Hard Tissues
At 6 months post-treatment, significant improvements were observed, particularly in patients with stage III periodontitis (AL ≥5 mm).
Attachment Loss (Primary Outcome)
In stage III patients:
DPSC group: 1.67 ± 1.508 mm improvement (26.81%)
Saline group: 1.03 ± 1.310 mm improvement (17.43%)
Statistically significant difference (P = 0.0338)
Probing Depth
DPSC group: 1.81 ± 1.490 mm reduction
Saline group: 1.08 ± 1.289 mm reduction
Significant improvement (P = 0.0147)
Bone Defect Depth (BDD)
Regeneration of alveolar bone remains one of the most difficult objectives in non-surgical periodontal therapy.
DPSC group: 0.24 ± 0.471 mm improvement
Saline group: 0.02 ± 0.348 mm improvement
Significant difference (P = 0.0147)
These findings demonstrate that DPSC injection promotes measurable alveolar bone regeneration—an outcome rarely achieved with non-surgical approaches.

Standard operating procedure for clinical DPSC injection (a) Representative image of the injection site. b Schematic diagram illustrating the DPSC injection procedure
Indications: Which Cases Benefit Most?
Post hoc analysis revealed important clinical insights:
Stage III Periodontitis
Patients with severe attachment loss showed significantly greater benefit compared to moderate cases.
Intrabony Defect Morphology
Narrow intrabony defects (<25°) responded more favorably
Flat defects (>25°) also improved, but to a lesser degree
Tooth Anatomy
Single-root teeth showed better outcomes than multi-root teeth
Multi-root teeth without furcation involvement demonstrated improved response
Furcation involvement reduced regenerative predictability
These findings help define optimal case selection for DPSC injection therapy.
Mechanism of Action: Beyond Cell Replacement
DPSC therapy promotes periodontal regeneration through multiple mechanisms:
Direct differentiation into osteoblast-like cells
Secretion of growth factors and cytokines
Immunomodulation of the inflammatory microenvironment
Activation of host stem cells
Restoration of periodontal tissue homeostasis
Rather than simply filling a defect, DPSCs recalibrate the molecular and cellular network necessary for organized tissue reconstruction.
Injection Technique: Minimally Invasive Protocol
The procedure is straightforward:
Complete SRP and mechanical debridement
Assess bone defect depth
Inject 0.6 mL DPSC suspension (1 × 10⁷ cells) into the defect
Overfill defect while withdrawing the needle
No flap elevation or surgical membrane placement is required.
Patients were monitored for 24 hours post-procedure and followed for 6 months.
Comparison to Surgical Regeneration
In previous animal studies using surgical stem cell implantation, attachment gain reached 2.9 ± 0.90 mm. In the current minimally invasive human trial, attachment gain reached 1.67 ± 1.51 mm.
Although slightly lower than surgical transplantation, DPSC injection offers:
Reduced patient morbidity
No surgical flap
Lower infection risk
Faster recovery
Greater patient acceptance
Reduced technical complexity
When compared to enamel matrix derivative therapy, clinical outcomes were comparable at the 6-month mark, though longer follow-up is required for definitive comparison.
Clinical Implications for Periodontists and General Dentists
DPSC injection represents a paradigm shift in periodontal regeneration:
Non-surgical alternative for stage III periodontitis
Potential to delay or eliminate the need for regenerative surgery
Improved patient comfort and acceptance
Regeneration of both attachment and bone
For clinicians managing advanced chronic periodontitis, this biologic therapy may soon expand the non-surgical armamentarium.
Limitations and Future Directions
While promising, several considerations remain:
Larger multicenter trials are required
Long-term follow-up beyond 6 months is necessary
Optimal dosing protocols must be refined
Cost-effectiveness analyses are needed
Immunogenicity risk requires continued evaluation
Future research will determine whether earlier-stage periodontitis may benefit from lower doses and whether repeated injections enhance outcomes.
Conclusion: A Breakthrough in Minimally Invasive Periodontal Regeneration
Dental pulp stem cell injection, combined with scaling and root planing, demonstrated significant improvement in attachment loss, probing depth, and alveolar bone regeneration in patients with chronic periodontitis—particularly those with stage III disease.
The therapy is safe, minimally invasive, and biologically sophisticated, restoring both soft and hard periodontal tissues through modulation of the local immune and regenerative environment.
As stem cell therapeutics continue to evolve, DPSC injection may redefine the standard of care for periodontal regeneration—shifting from surgical reconstruction toward biologically driven, minimally invasive regeneration.
The future of periodontal disease treatment is regenerative, and dental stem cell therapy is leading that transformation.
Frequently Asked Questions (FAQ)
1. What is dental pulp stem cell (DPSC) injection therapy?
Dental pulp stem cell (DPSC) injection therapy is a minimally invasive regenerative treatment for periodontitis. It involves injecting mesenchymal stem cells derived from dental pulp directly into periodontal intrabony defects following scaling and root planing (SRP), with the goal of restoring attachment and promoting alveolar bone regeneration—without surgical flap elevation.
2. How does DPSC injection differ from traditional periodontal regenerative surgery?
Conventional regenerative procedures such as guided tissue regeneration (GTR), bone grafting, and enamel matrix derivatives require surgical flap elevation, membrane placement, and grafting materials. In contrast, DPSC injection therapy is delivered via a localized injection into the defect site, reducing patient morbidity, postoperative discomfort, infection risk, and surgical complexity.
3. Is DPSC injection therapy safe?
In randomized, placebo-controlled clinical trials involving 132 patients, DPSC injection therapy demonstrated a favorable safety profile. No serious adverse events were reported. Mild events such as transient toothache or gingival swelling were self-limiting and resolved without intervention. Cell preparations showed high viability and no microbial contamination.
4. What clinical improvements were observed with DPSC therapy?
At six months post-treatment, patients receiving DPSC injections showed statistically significant improvements in:
Attachment loss (AL)
Probing depth (PD)
Alveolar bone defect depth (BDD)
The most pronounced benefits were observed in stage III periodontitis cases, particularly in sites with intrabony defects.
5. Which patients benefit most from dental stem cell injection therapy?
Data suggest the strongest outcomes occur in:
Stage III periodontitis (≥5 mm attachment loss)
Narrow intrabony defects (<25° defect angle)
Single-root teeth
Multi-root teeth without furcation involvement
Furcation involvement may reduce regenerative predictability.
6. How do dental pulp stem cells promote periodontal regeneration?
DPSCs contribute to regeneration through multiple mechanisms:
Differentiation into osteoblast-like cells
Secretion of growth factors and cytokines
Immunomodulation of the inflammatory microenvironment
Activation of host regenerative pathways
Rather than merely filling defects, DPSCs help restore periodontal tissue homeostasis and support coordinated regeneration of bone, ligament, and connective tissue.
7. Does DPSC therapy eliminate the need for conventional scaling and root planing?
No. Standard periodontal therapy, including oral hygiene instruction and scaling and root planing (SRP), is performed prior to stem cell injection. DPSC therapy serves as a regenerative adjunct, enhancing healing and tissue reconstruction beyond what SRP alone typically achieves in advanced cases.
8. Is DPSC injection currently a replacement for surgical regeneration?
While results are promising, DPSC injection is not yet a universal replacement for surgical techniques. Larger multicenter trials and long-term follow-up studies are needed. However, it represents a significant step toward minimally invasive biologic periodontal regeneration, particularly for stage III disease.
9- Is there any commercial DSPC injection available for sale?
Reference:
Impact of allogeneic dental pulp stem cell injection on tissue regeneration in periodontitis: a multicenter randomized clinical trial
Yi Liu 1,2,3,✉,#, Yitong Liu 1,#, Jingchao Hu 1,#, Jianhui Han 4, Lin Song 1, Xu Liu 1, Nannan Han 1, Xia Xia 5,#, Jia He 5, Hongfang Meng 5, Meng Wan 6,#, Haojie Wang 6, Xiaodan Liu 6, Zhanyi Gao 6, Xiao Wang 6,✉, Chutse Wu 5,✉, Songlin Wang 2,3,7,8,9,✉
Recent Articles
In our previous article, we examined emerging evidence linking periodontal pathogens to systemic disease, including their potential role in breast cancer progression. That discussion underscored a critical reality: periodontal disease is not merely a localized oral condition, but a chronic inflammatory disease with far-reaching biological implications.
Affecting more than 700 million people worldwide, periodontal disease remains one of the leading causes of tooth loss in adults. It is characterized by progressive destruction of the alveolar bone, periodontal ligament, and connective tissue attachment apparatus. In advanced cases—particularly stage III disease—management of intrabony defects and attachment loss presents a substantial clinical challenge.
Conventional regenerative approaches, such as guided tissue regeneration (GTR), bone grafting, and the use of biologic mediators, including growth factors or enamel matrix derivatives, rely on invasive surgical protocols. While effective in selected cases, these procedures are associated with postoperative morbidity, patient discomfort, infection risk, and inconsistent regenerative outcomes.
In this article, we explore one of the most innovative developments in periodontal therapy based on recent clinical research: dental pulp stem cell (DPSC) injection therapy. This minimally invasive, biologically driven approach is designed to restore periodontal microenvironment homeostasis and stimulate regeneration of both soft and hard tissues—without the need for surgical intervention.
Understanding Periodontal Disease and the Need for Regenerative Innovation
Periodontitis is driven by dysbiotic biofilms and a dysregulated host immune response. The resulting chronic inflammation leads to:
Clinical attachment loss (AL)
Increased periodontal probing depth (PD)
Gingival recession (GR)
Tooth mobility (TM)
Intrabony defects and alveolar bone resorption
While scaling and root planing (SRP) effectively reduces microbial burden, it does not reliably regenerate lost bone or periodontal attachment in moderate-to-severe cases. Surgical regenerative techniques attempt to address this limitation but often yield incomplete or unpredictable tissue reconstruction.
The complexity of periodontal regeneration lies in restoring a coordinated architecture consisting of cementum, periodontal ligament, and alveolar bone. Achieving this requires more than mechanical debridement—it requires biologic modulation.
Why Dental Pulp Stem Cells?
Dental pulp stem cells (DPSCs) are mesenchymal stem cells derived from extracted third molars or orthodontically indicated teeth. Compared with bone marrow–derived or adipose-derived mesenchymal stem cells, DPSCs offer several advantages:
Minimally invasive harvesting
High proliferative capacity
Strong osteogenic differentiation potential
Resistance to inflammation-induced senescence
Low immunogenicity
Immunomodulatory properties
DPSCs express canonical mesenchymal markers CD73, CD90, and CD105, while lacking hematopoietic and immune lineage markers such as CD19, CD34, CD45, CD11b, and HLA-DR. They demonstrate robust osteogenic gene expression, including ALP, RUNX2, BMP2, OPN, and OSX.
Beyond differentiation, DPSCs actively regulate the immune microenvironment. They modulate T cells, B cells, macrophages, dendritic cells, and neutrophils, contributing to the restoration of periodontal microenvironment balance—an essential prerequisite for tissue regeneration.
First-in-Human Randomized Clinical Trials of DPSC Injection
To evaluate safety and efficacy, two randomized, placebo-controlled clinical trials were conducted in China, enrolling 132 patients with chronic periodontitis (158 treated teeth).
Study Design Overview
Total participants: 132
Centers: Two clinical institutions
Primary endpoint: Attachment loss (AL) at 6 months
Secondary endpoints: PD, GR, TM, bone defect depth (BDD)
Intervention: DPSC injection vs saline placebo
Patients received standard periodontal therapy, including oral hygiene instruction and mechanical debridement prior to injection.
In the investigator-initiated trial:
Single injection group: 1 × 10⁷ DPSCs
Double injection group: 1 × 10⁷ DPSCs × 2 (30-day interval)
Saline control group
In the Phase I dose-escalation trial:
Doses ranged from 1 × 10⁶ to 1 × 10⁷ DPSCs per tooth
All injections were delivered directly into the intrabony defect using a minimally invasive technique following SRP.
Safety Profile: Favorable and Well Tolerated
A critical barrier to stem cell therapy adoption is safety. Across both trials:
No serious adverse events were reported
Mild adverse events were limited to grade 1 (toothache, gingival swelling, mild diarrhea)
All adverse effects resolved spontaneously without intervention
Cell preparations demonstrated:
90% viability
Absence of bacterial contamination
Minimal residual processing enzymes
The findings confirm that allogeneic DPSC injection is safe and well-tolerated in the treatment of periodontal disease.

DPSC injection demonstrated greater efficacy in bone defect regeneration compared to saline injection. a, c In narrow initial bone defects, CBCT scans show that DPSC injection significantly reduced the alveolar bone resorption height at day 180 post-treatment. Notably, DPSC injection also reduced signs of periodontitis-induced maxillary sinus inflammation in some cases, as indicated by the yellow dotted line in (c), providing further evidence of its therapeutic effects on local inflammation. b, d In flat initial bone defects, CBCT scans also revealed therapeutic benefits of DPSC injection, although to a lesser extent compared with the narrow defect group. The yellow stars indicate the chief complaint tooth, and the yellow arrows indicate the bone defect
Clinical Efficacy: Regeneration of Both Soft and Hard Tissues
At 6 months post-treatment, significant improvements were observed, particularly in patients with stage III periodontitis (AL ≥5 mm).
Attachment Loss (Primary Outcome)
In stage III patients:
DPSC group: 1.67 ± 1.508 mm improvement (26.81%)
Saline group: 1.03 ± 1.310 mm improvement (17.43%)
Statistically significant difference (P = 0.0338)
Probing Depth
DPSC group: 1.81 ± 1.490 mm reduction
Saline group: 1.08 ± 1.289 mm reduction
Significant improvement (P = 0.0147)
Bone Defect Depth (BDD)
Regeneration of alveolar bone remains one of the most difficult objectives in non-surgical periodontal therapy.
DPSC group: 0.24 ± 0.471 mm improvement
Saline group: 0.02 ± 0.348 mm improvement
Significant difference (P = 0.0147)
These findings demonstrate that DPSC injection promotes measurable alveolar bone regeneration—an outcome rarely achieved with non-surgical approaches.

Standard operating procedure for clinical DPSC injection (a) Representative image of the injection site. b Schematic diagram illustrating the DPSC injection procedure
Indications: Which Cases Benefit Most?
Post hoc analysis revealed important clinical insights:
Stage III Periodontitis
Patients with severe attachment loss showed significantly greater benefit compared to moderate cases.
Intrabony Defect Morphology
Narrow intrabony defects (<25°) responded more favorably
Flat defects (>25°) also improved, but to a lesser degree
Tooth Anatomy
Single-root teeth showed better outcomes than multi-root teeth
Multi-root teeth without furcation involvement demonstrated improved response
Furcation involvement reduced regenerative predictability
These findings help define optimal case selection for DPSC injection therapy.
Mechanism of Action: Beyond Cell Replacement
DPSC therapy promotes periodontal regeneration through multiple mechanisms:
Direct differentiation into osteoblast-like cells
Secretion of growth factors and cytokines
Immunomodulation of the inflammatory microenvironment
Activation of host stem cells
Restoration of periodontal tissue homeostasis
Rather than simply filling a defect, DPSCs recalibrate the molecular and cellular network necessary for organized tissue reconstruction.
Injection Technique: Minimally Invasive Protocol
The procedure is straightforward:
Complete SRP and mechanical debridement
Assess bone defect depth
Inject 0.6 mL DPSC suspension (1 × 10⁷ cells) into the defect
Overfill defect while withdrawing the needle
No flap elevation or surgical membrane placement is required.
Patients were monitored for 24 hours post-procedure and followed for 6 months.
Comparison to Surgical Regeneration
In previous animal studies using surgical stem cell implantation, attachment gain reached 2.9 ± 0.90 mm. In the current minimally invasive human trial, attachment gain reached 1.67 ± 1.51 mm.
Although slightly lower than surgical transplantation, DPSC injection offers:
Reduced patient morbidity
No surgical flap
Lower infection risk
Faster recovery
Greater patient acceptance
Reduced technical complexity
When compared to enamel matrix derivative therapy, clinical outcomes were comparable at the 6-month mark, though longer follow-up is required for definitive comparison.
Clinical Implications for Periodontists and General Dentists
DPSC injection represents a paradigm shift in periodontal regeneration:
Non-surgical alternative for stage III periodontitis
Potential to delay or eliminate the need for regenerative surgery
Improved patient comfort and acceptance
Regeneration of both attachment and bone
For clinicians managing advanced chronic periodontitis, this biologic therapy may soon expand the non-surgical armamentarium.
Limitations and Future Directions
While promising, several considerations remain:
Larger multicenter trials are required
Long-term follow-up beyond 6 months is necessary
Optimal dosing protocols must be refined
Cost-effectiveness analyses are needed
Immunogenicity risk requires continued evaluation
Future research will determine whether earlier-stage periodontitis may benefit from lower doses and whether repeated injections enhance outcomes.
Conclusion: A Breakthrough in Minimally Invasive Periodontal Regeneration
Dental pulp stem cell injection, combined with scaling and root planing, demonstrated significant improvement in attachment loss, probing depth, and alveolar bone regeneration in patients with chronic periodontitis—particularly those with stage III disease.
The therapy is safe, minimally invasive, and biologically sophisticated, restoring both soft and hard periodontal tissues through modulation of the local immune and regenerative environment.
As stem cell therapeutics continue to evolve, DPSC injection may redefine the standard of care for periodontal regeneration—shifting from surgical reconstruction toward biologically driven, minimally invasive regeneration.
The future of periodontal disease treatment is regenerative, and dental stem cell therapy is leading that transformation.
Frequently Asked Questions (FAQ)
1. What is dental pulp stem cell (DPSC) injection therapy?
Dental pulp stem cell (DPSC) injection therapy is a minimally invasive regenerative treatment for periodontitis. It involves injecting mesenchymal stem cells derived from dental pulp directly into periodontal intrabony defects following scaling and root planing (SRP), with the goal of restoring attachment and promoting alveolar bone regeneration—without surgical flap elevation.
2. How does DPSC injection differ from traditional periodontal regenerative surgery?
Conventional regenerative procedures such as guided tissue regeneration (GTR), bone grafting, and enamel matrix derivatives require surgical flap elevation, membrane placement, and grafting materials. In contrast, DPSC injection therapy is delivered via a localized injection into the defect site, reducing patient morbidity, postoperative discomfort, infection risk, and surgical complexity.
3. Is DPSC injection therapy safe?
In randomized, placebo-controlled clinical trials involving 132 patients, DPSC injection therapy demonstrated a favorable safety profile. No serious adverse events were reported. Mild events such as transient toothache or gingival swelling were self-limiting and resolved without intervention. Cell preparations showed high viability and no microbial contamination.
4. What clinical improvements were observed with DPSC therapy?
At six months post-treatment, patients receiving DPSC injections showed statistically significant improvements in:
Attachment loss (AL)
Probing depth (PD)
Alveolar bone defect depth (BDD)
The most pronounced benefits were observed in stage III periodontitis cases, particularly in sites with intrabony defects.
5. Which patients benefit most from dental stem cell injection therapy?
Data suggest the strongest outcomes occur in:
Stage III periodontitis (≥5 mm attachment loss)
Narrow intrabony defects (<25° defect angle)
Single-root teeth
Multi-root teeth without furcation involvement
Furcation involvement may reduce regenerative predictability.
6. How do dental pulp stem cells promote periodontal regeneration?
DPSCs contribute to regeneration through multiple mechanisms:
Differentiation into osteoblast-like cells
Secretion of growth factors and cytokines
Immunomodulation of the inflammatory microenvironment
Activation of host regenerative pathways
Rather than merely filling defects, DPSCs help restore periodontal tissue homeostasis and support coordinated regeneration of bone, ligament, and connective tissue.
7. Does DPSC therapy eliminate the need for conventional scaling and root planing?
No. Standard periodontal therapy, including oral hygiene instruction and scaling and root planing (SRP), is performed prior to stem cell injection. DPSC therapy serves as a regenerative adjunct, enhancing healing and tissue reconstruction beyond what SRP alone typically achieves in advanced cases.
8. Is DPSC injection currently a replacement for surgical regeneration?
While results are promising, DPSC injection is not yet a universal replacement for surgical techniques. Larger multicenter trials and long-term follow-up studies are needed. However, it represents a significant step toward minimally invasive biologic periodontal regeneration, particularly for stage III disease.
9- Is there any commercial DSPC injection available for sale?
Reference:
Impact of allogeneic dental pulp stem cell injection on tissue regeneration in periodontitis: a multicenter randomized clinical trial
Yi Liu 1,2,3,✉,#, Yitong Liu 1,#, Jingchao Hu 1,#, Jianhui Han 4, Lin Song 1, Xu Liu 1, Nannan Han 1, Xia Xia 5,#, Jia He 5, Hongfang Meng 5, Meng Wan 6,#, Haojie Wang 6, Xiaodan Liu 6, Zhanyi Gao 6, Xiao Wang 6,✉, Chutse Wu 5,✉, Songlin Wang 2,3,7,8,9,✉
Recent Articles
Pulsed Electromagnetic Field (PEMF) Use in Implant Dentistry
Pulsed Electromagnetic Field (PEMF) Use in Implant Dentistry Abstract and Historical Background Pulsed Electromagnetic Field (PEMF) therapy refers to the application of low-frequency electromagnetic [...]
Laser Therapy in the Management of Peri-Implantitis
Laser Therapy in the Management of Peri-Implantitis Laser technology has become an increasingly valuable adjunct in the treatment of peri-implant diseases, particularly peri-implantitis. Its [...]
Magnetically Guided Dental Nanobots (CalBots) Are Real
Magnetically Guided Dental Nanobots (CalBots) Are Real The concept of nanobots in dentistry has long been associated with futuristic speculation. However, recent peer-reviewed research [...]
Peri-Implantitis Treatment & Prevention Methods
Peri-Implantitis Treatment & Prevention Methods Peri-implantitis remains one of the most significant biological complications affecting dental implants, posing a serious threat to long-term implant [...]
Latest Sinus Lifting Techniques in Modern Implant Dentistry
Latest Sinus Lifting Techniques in Modern Implant Dentistry A Comprehensive Clinical Review Introduction to Sinus Lifting in Implantology Sinus lifting, or maxillary sinus floor [...]
Biomechanics of the Triple Abutment & BOPiT Concept
Dental Biomechanics · Implant Science · Clinical Evidence Biomechanics of the Triple Abutment & BOPiT Concept How a saddle-shaped mathematical surface is rewriting the rules of load distribution [...]